The Fish Oil Paradox : When the Evidence Refuses to Cooperate
People have been recommending fish oil capsules with the confidence of prescribing aspirin. The mechanistic story seemed ironclad: omega-3 fatty acids lower triglycerides, reduce inflammation, stabilize atherosclerotic plaques, and decrease platelet aggregation. Patients dutifully swallowed billions of capsules annually, trusting that their hearts would thank them. The evidence, however, tells a more humbling story.
The most comprehensive systematic assessment to date—a 2020 Cochrane meta-analysis encompassing 86 randomized trials and over 162,000 participants—delivers a sobering conclusion. According to this analysis, increasing intake of long-chain omega-3 fatty acids (EPA and DHA) shows little or no effect on all-cause mortality (RR 0.97, 95% CI 0.93–1.01) or total cardiovascular events (RR 0.96, 95% CI 0.92–1.01) based on high-certainty evidence (Abdelhamid et al., Cochrane 2020). The data suggest possible modest reductions in coronary heart disease events (NNT 167) and coronary mortality (NNT 334), but these effects carry only low-certainty evidence and evaporate in sensitivity analyses restricted to low-bias trials.
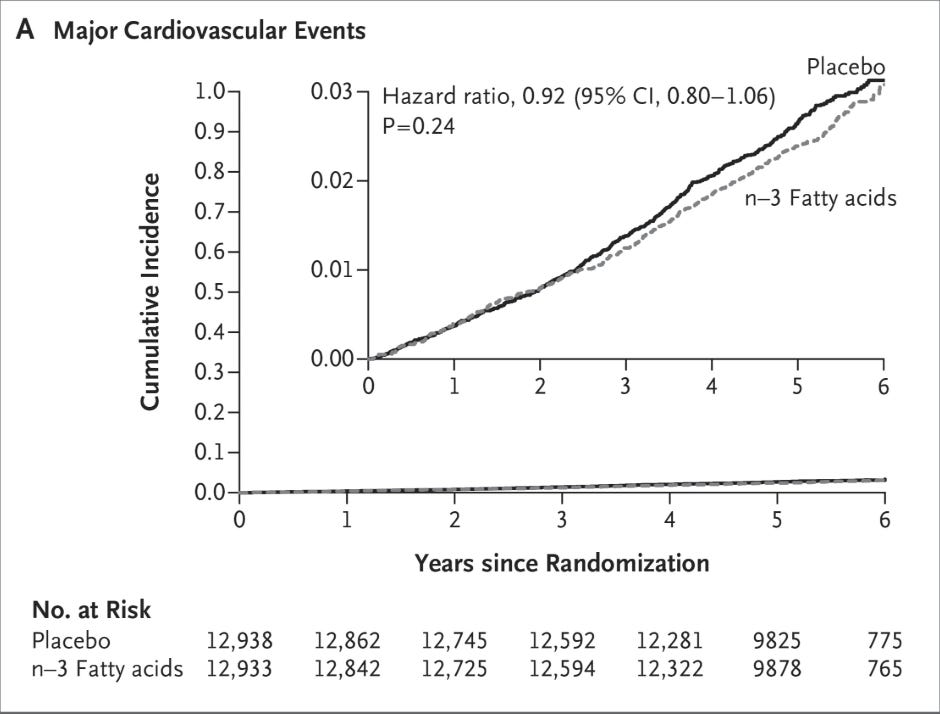
The VITAL trial reinforced this disappointment. In nearly 26,000 adults without prior cardiovascular disease, standard-dose marine omega-3 supplementation (1 g/day) over 5.3 years produced no significant reduction in the primary composite endpoint compared to placebo (HR 0.92, 95% CI 0.80–1.06). However, a notable 28% reduction in myocardial infarction was observed (HR 0.72, 95% CI 0.59–0.90)—a secondary endpoint that complicates any simple dismissal (Manson et al., NEJM 2019).
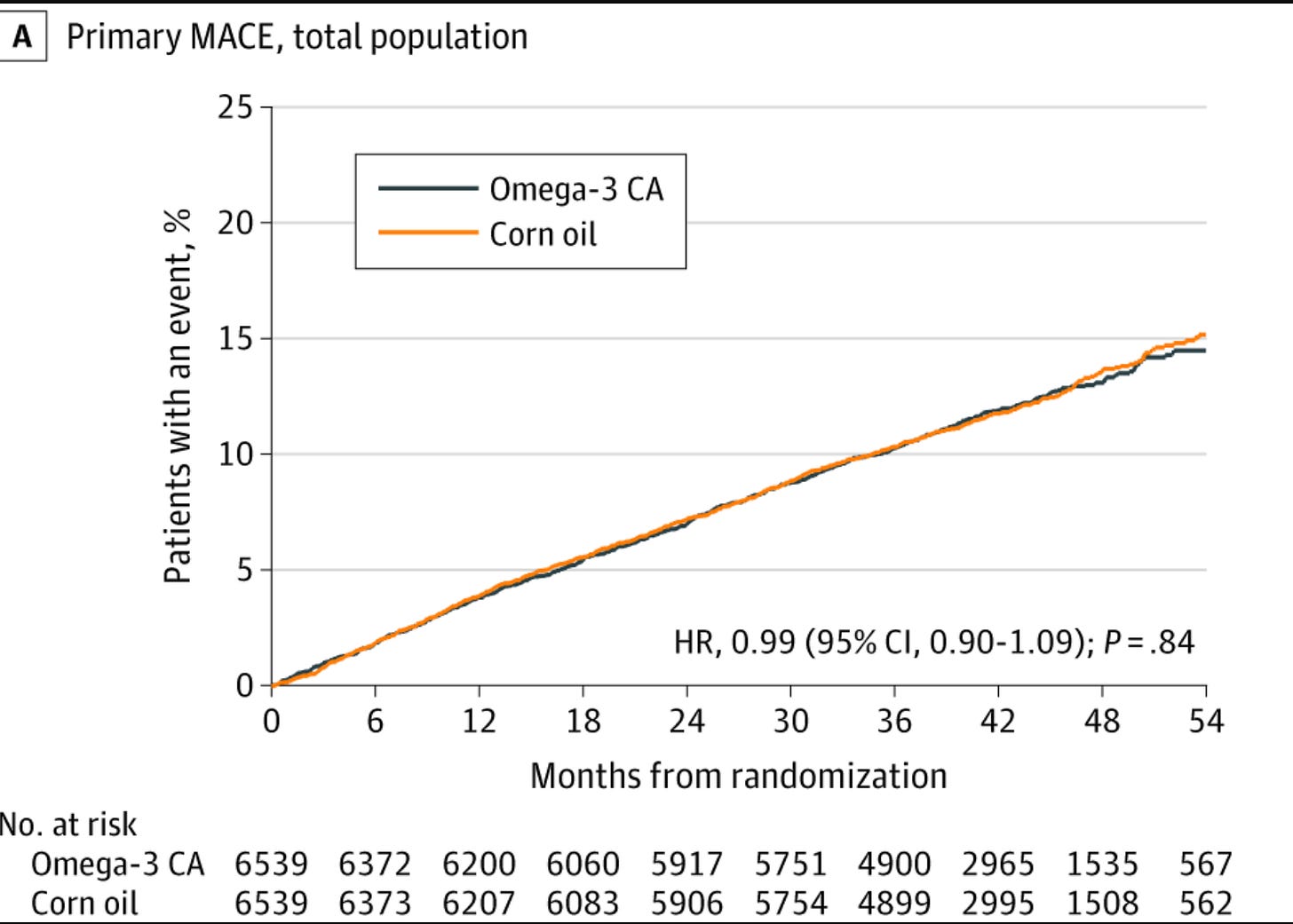
The STRENGTH trial delivered similarly null results: among 13,078 high-risk patients with atherogenic dyslipidemia, a carboxylic acid formulation of EPA plus DHA showed no benefit over corn oil for major adverse cardiovascular events (HR 0.99, 95% CI 0.90–1.09) (Nicholls et al., JAMA 2020).
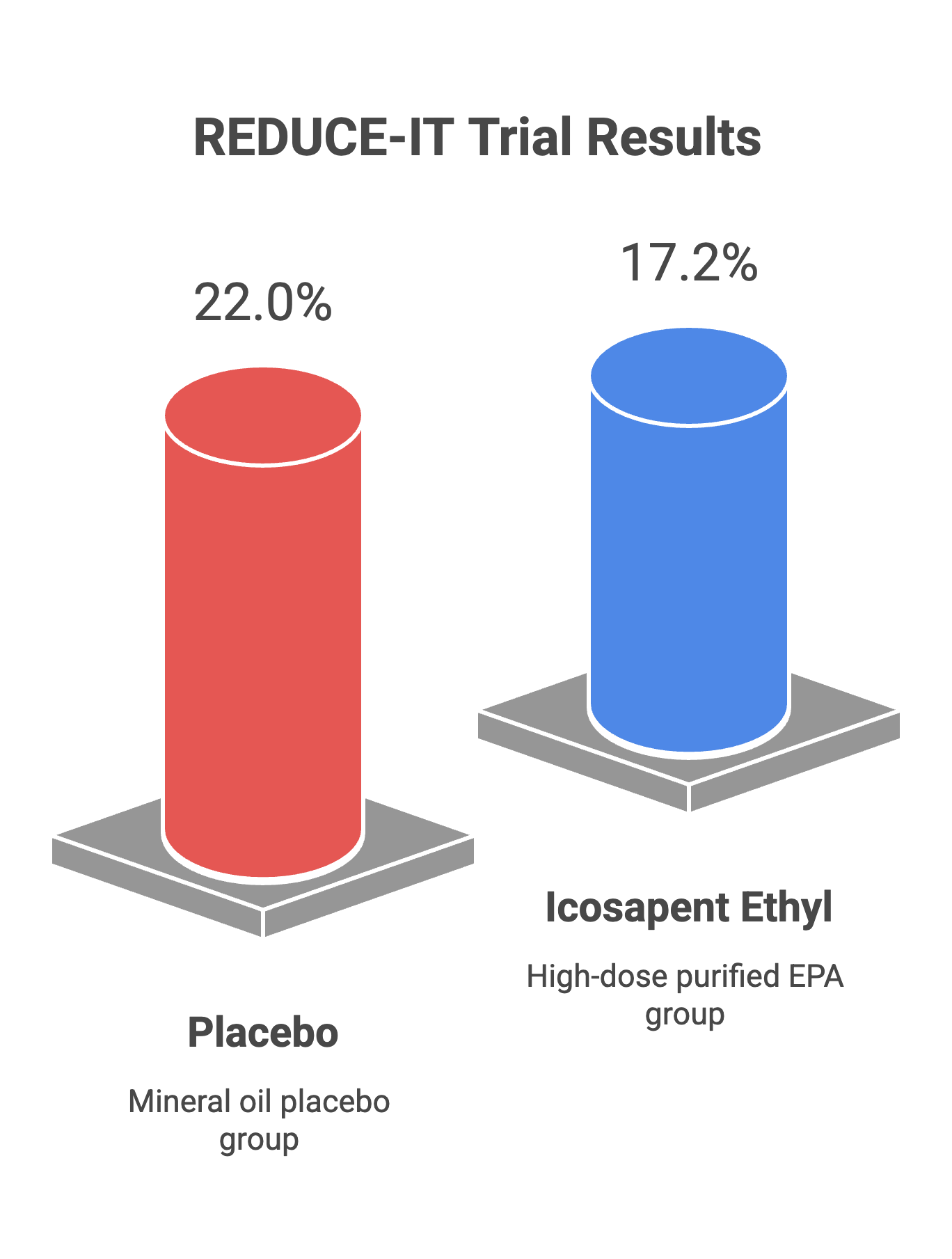
Yet one trial defied this pattern. REDUCE-IT, funded by Amarin Pharma, randomized 8,179 patients with established cardiovascular disease or diabetes to high-dose icosapent ethyl (4 g/day of purified EPA) versus mineral oil placebo. The results were striking: a 25% relative reduction in the primary composite endpoint (HR 0.75, 95% CI 0.68–0.83), corresponding to an absolute risk reduction of 4.8 percentage points (22.0% vs 17.2%) and a number needed to treat of approximately 21 over 4.9 years. Cardiovascular death was also reduced by 20% (Bhatt et al., NEJM 2019). An updated meta-analysis confirmed dose-response relationships for cardiovascular outcomes (Hu et al., JAHA 2019).
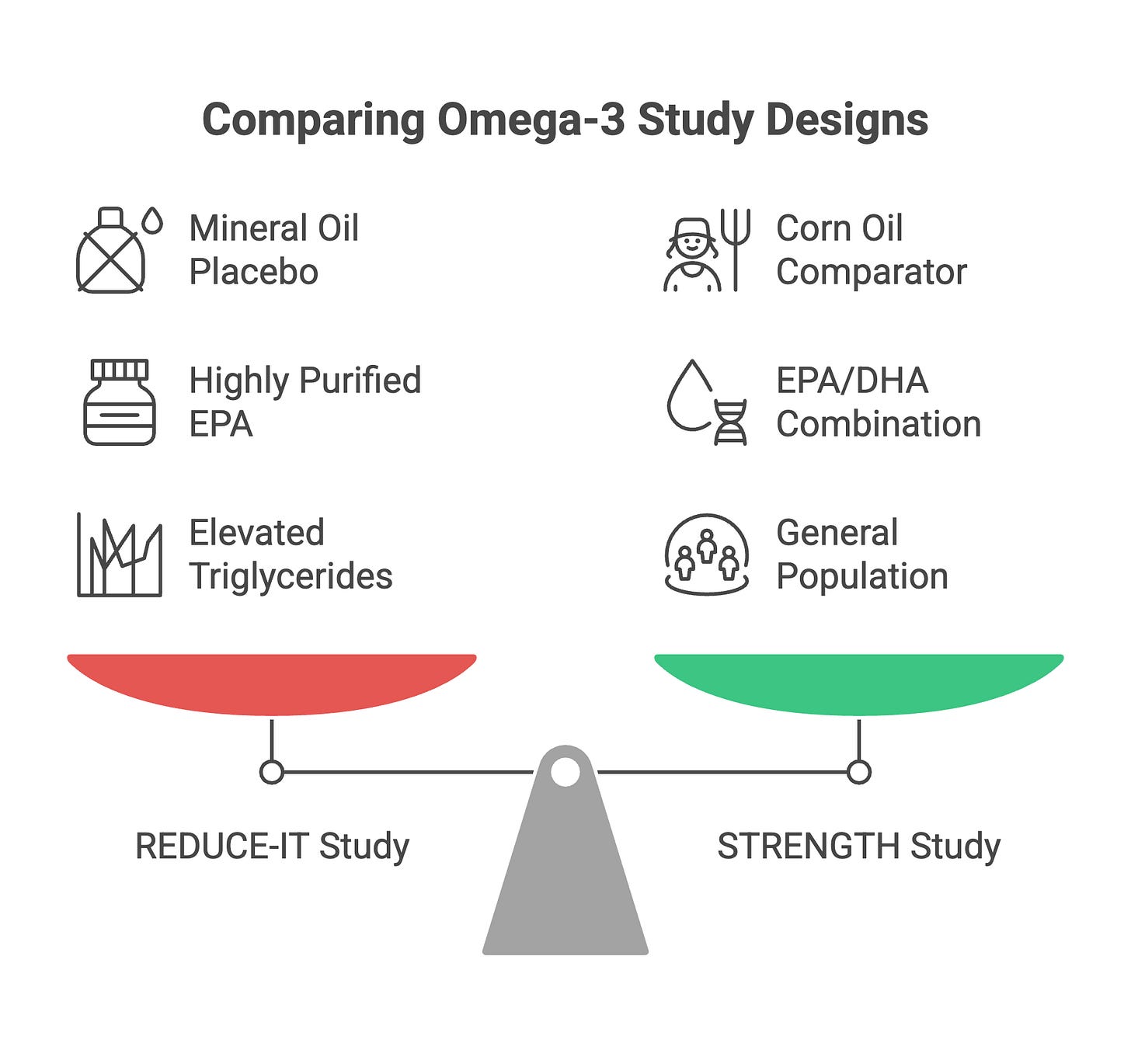
How do we reconcile these findings? Both REDUCE-IT and STRENGTH face legitimate methodological questions. The mineral oil placebo in REDUCE-IT has drawn criticism for potentially raising LDL and inflammatory markers, which could inflate apparent benefit. Conversely, STRENGTH’s corn oil comparator contains linoleic acid—itself potentially cardioprotective—possibly obscuring any omega-3 effect. Neither comparator was truly inert.
The answer may lie not in whether omega-3s work, but in which formulation, at what dose, and in whom. REDUCE-IT used highly purified EPA at pharmacological doses in patients with elevated triglycerides—a distinct intervention from over-the-counter fish oil capsules in the general population. Recent imaging data suggest EPA, but not EPA/DHA combinations, reduces coronary plaque burden (Sheppard et al., Acta Cardiol 2025). Whether EPA’s apparent benefit stems from triglyceride lowering, anti-inflammatory effects, membrane stabilization, or mechanisms yet unidentified remains uncertain. The increased atrial fibrillation observed with icosapent ethyl (3.1% vs 2.1%) also warrants consideration.
For now, the message is nuanced: generic fish oil supplements for primary prevention lack convincing evidence of benefit. High-dose prescription icosapent ethyl may reduce cardiovascular events in select high-risk patients with elevated triglycerides despite statin therapy—though at the cost of increased arrhythmia risk. Between these poles lies considerable uncertainty, and a reminder that biological plausibility cannot substitute for rigorous trial data.
If you liked what you read share it.