Sudden Cardiac Death in Bodybuilders: What the Data Shows
Five deaths in a year—including some as young as 19. Here’s what the evidence reveals about the intersection of performance-enhancing substances, silent heart conditions, and sudden death in the gym.
Last month, the Economic Times ran a headline that caught my attention: “Varinder Singh Ghuman’s death: Five bodybuilders, some as young as 19, died in a year.”
Ghuman was a bodybuilding icon—a 40-year-old who had built his physique over decades. Within the same year, five other bodybuilders died suddenly, some barely out of their teens. The news went viral. Your WhatsApp groups lit up with theories: COVID vaccines, whey protein, “overexercising.” Every forward had a different culprit.
I get it. When young, seemingly invincible athletes drop dead, we want a simple villain to blame. The recently published European Heart Journal study on bodybuilding mortality gives us the clearest answer we’ve had.
I’m going to walk through the study’s findings, explain the cardiac conditions that make athletes vulnerable, identify the substances that turn muscle-building into heart damage, and show what this means for screening and prevention. The evidence tells a very different story than the one circulating on social media—and it’s both more specific and more actionable.
1. The bodybuilding mortality study
What the autopsy data actually shows
A 2025 study in the European Heart Journal tracked 20,286 male bodybuilders over 8.1 years—190,211 athlete-years of follow-up.[1] During this period, 121 athletes died. Forty-six of these deaths were sudden cardiac deaths, accounting for 38% of all mortality in this cohort.
The numbers are striking. Among currently competing bodybuilders, the sudden cardiac death incidence was 32.83 per 100,000 athlete-years. Professional bodybuilders faced a 5.23-fold higher risk than amateur competitors. The average age at sudden cardiac death was 34.7 years for those actively competing, though the overall average across all deaths was 42.2 years.
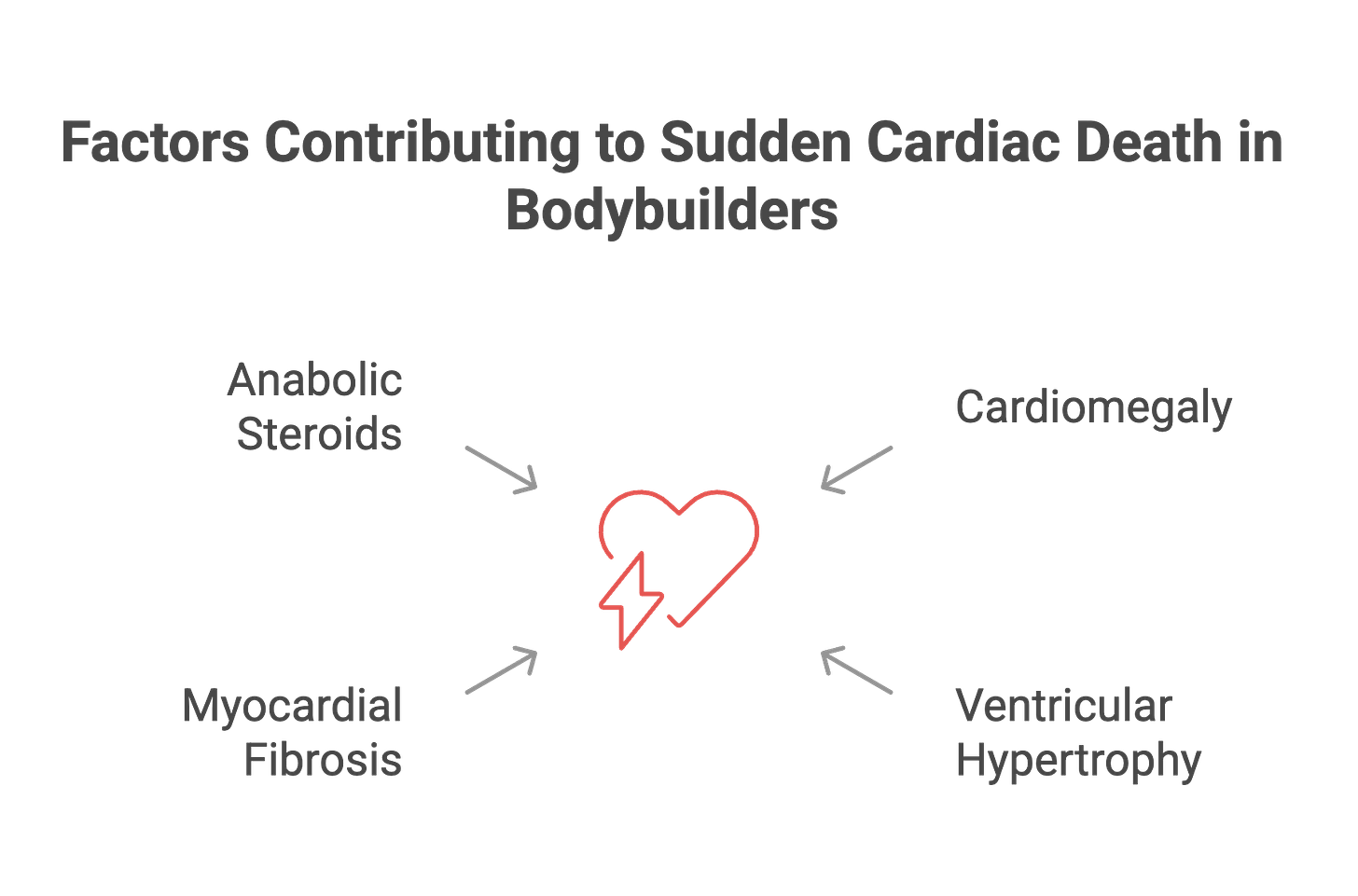
The autopsy findings revealed what was happening inside these hearts. Cardiomegaly—enlarged hearts—was universal. The hearts weighed 73.7% more than normal reference values. Severe ventricular hypertrophy was present in every case. Myocardial fibrosis—scar tissue replacing normal heart muscle—created the substrate for fatal arrhythmias.
Toxicology reports detected anabolic-androgenic steroids in three of five tested cases. At least 16 athletes in the cohort had documented histories of performance-enhancing drug use. The pattern was consistent: structural heart disease, substance exposure, and sudden death during or shortly after intense physical exertion.
The study documented non-cardiac deaths too—suicides, homicides, overdoses, kidney failure. The mental health and substance abuse patterns extended beyond cardiovascular pathology, but that’s a separate discussion.
These numbers are stark. But they don’t tell us why these hearts failed. For that, we need to understand the underlying cardiac conditions that make sudden death possible.
2. The silent cardiac conditions
HCM, ARVC, and the diseases you can’t see coming
Sudden cardiac death in athletes typically results from conditions that remain clinically silent until triggered by exertion. The substrates for these events fall into distinct categories, and understanding them explains why these deaths occur without warning.
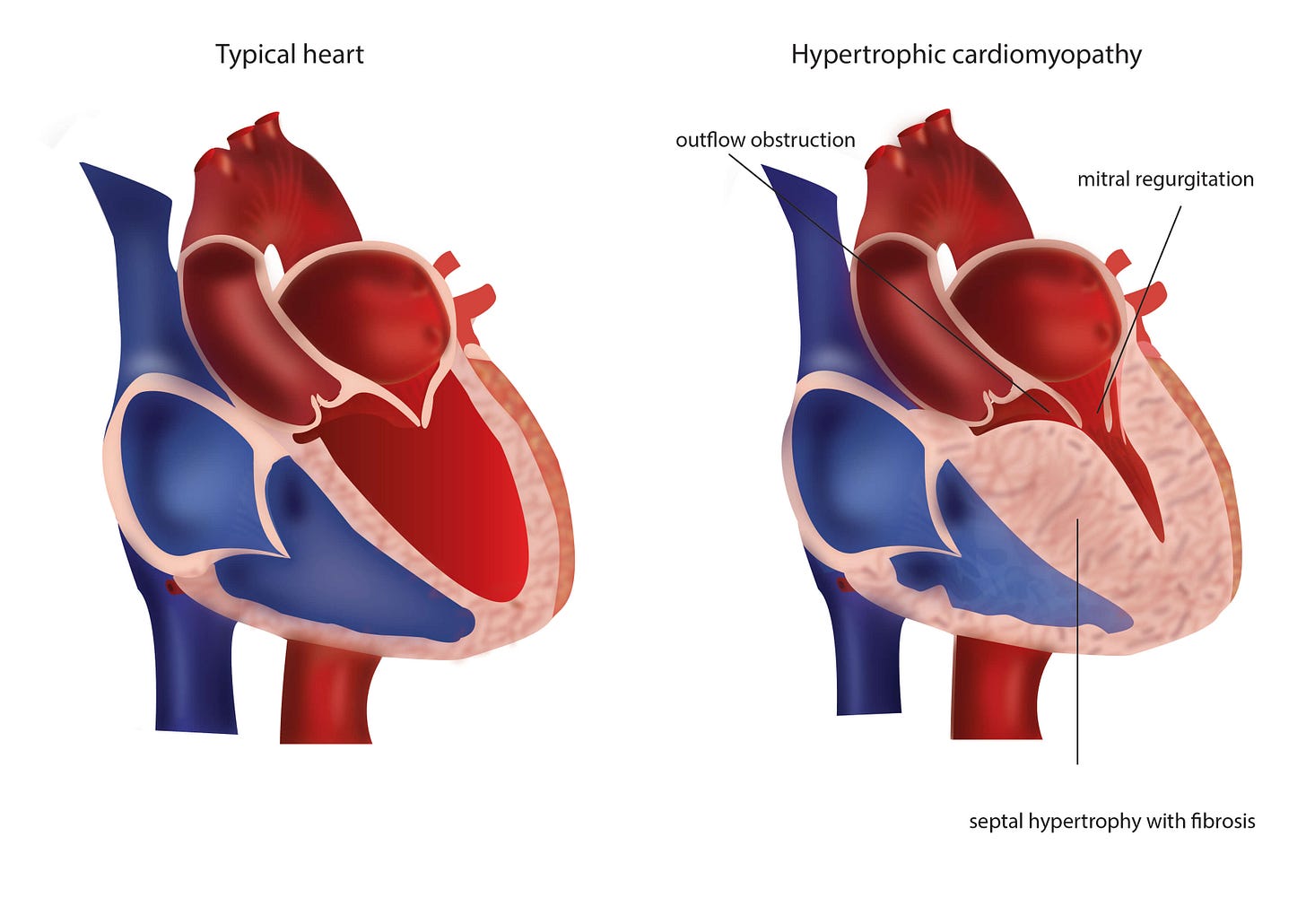
Hypertrophic cardiomyopathy (HCM) is the leading cause of sudden cardiac death in athletes under 35.[2] The disease is characterized by asymmetric left ventricular hypertrophy—the ventricular septum thickens disproportionately, creating both mechanical obstruction to blood flow and electrical instability.
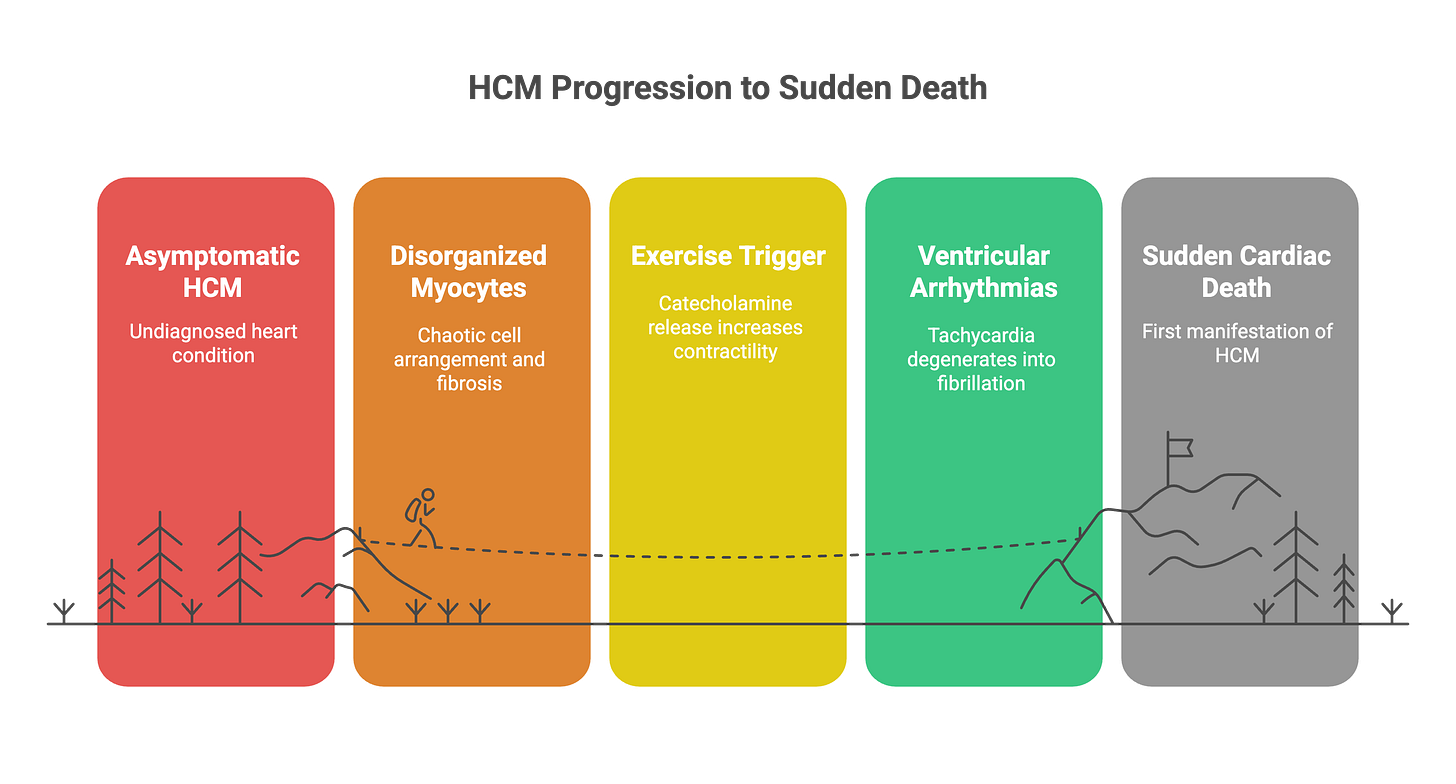
The myocardial architecture in HCM is disorganized. Instead of parallel myocyte alignment, the cells arrange in chaotic patterns with increased interstitial fibrosis. This creates the substrate for re-entrant ventricular arrhythmias. During intense exercise, catecholamine release increases myocardial contractility and heart rate. In HCM hearts, this heightened contractile state combined with the disorganized tissue architecture can trigger ventricular tachycardia that degenerates rapidly into ventricular fibrillation.
The clinical challenge is that HCM often presents asymptomatically. Studies show that sudden death is the first manifestation in approximately 30% of cases. The athlete feels fine, performs well, and has no prior symptoms. The diagnosis is missed until autopsy.
Arrhythmogenic right ventricular cardiomyopathy (ARVC) follows a different pathophysiology but produces similar outcomes. The disease involves progressive replacement of right ventricular myocardium with fibrofatty tissue. This replacement creates an arrhythmogenic substrate—the fibrotic regions form electrical barriers that promote re-entrant circuits.
ARVC has a paradoxical relationship with exercise. Mechanical stress from training accelerates myocyte death and fibrofatty replacement. Endurance athletes show higher prevalence, but the repetitive Valsalva maneuvers during heavy resistance training can impose significant right ventricular wall stress. Each training session potentially worsens the underlying pathology.
The right ventricle is technically difficult to image with standard echocardiography, which means ARVC is frequently underdiagnosed until advanced stages. Cardiac MRI has better sensitivity for detecting early fibrofatty changes, but this isn’t part of routine screening.
Ion channelopathies—Long QT syndrome, Brugada syndrome, catecholaminergic polymorphic ventricular tachycardia (CPVT)—are genetically determined electrical diseases. The structural heart appears normal on imaging. The problem is in the ion channels that control the cardiac action potential. These conditions are silent at rest. Exertion, stress, and catecholamine surges unmask them. A sprinting finish or a max-effort lift can trigger Torsades de Pointes or polymorphic ventricular tachycardia.
Coronary anomalies, particularly anomalous origin of the left coronary artery from the right sinus of Valsalva, cause sudden death through acute ischemia. During exertion, aortic root dilation compresses the anomalous vessel as it courses between the aorta and pulmonary artery. The resulting myocardial ischemia can trigger ventricular fibrillation. These anomalies are undetectable on ECG or standard echocardiography and require CT or MR angiography for diagnosis.
In athletes over 35, coronary artery disease becomes the dominant substrate.[3]
The bodybuilding mortality study showed the average age of sudden cardiac death was 34.7 years in actively competing athletes but 42.2 years overall—suggesting a transition point where atherosclerotic disease begins to contribute alongside cardiomyopathies.
Varinder Singh Ghuman was over 40. His death could have resulted from longstanding HCM that remained undiagnosed, ARVC accelerated by decades of training, or coronary artery disease compounded by other risk factors.
But the bodybuilding mortality study’s autopsy findings—hearts weighing 73.7% more than normal, severe hypertrophy, extensive fibrosis—suggest that these structural abnormalities weren’t purely from genetic cardiomyopathies. The degree of pathological remodeling points to an additional factor: anabolic-androgenic steroids.
3. The substance cascade
Anabolic steroids, pre-workouts, and the arrhythmogenic perfect storm
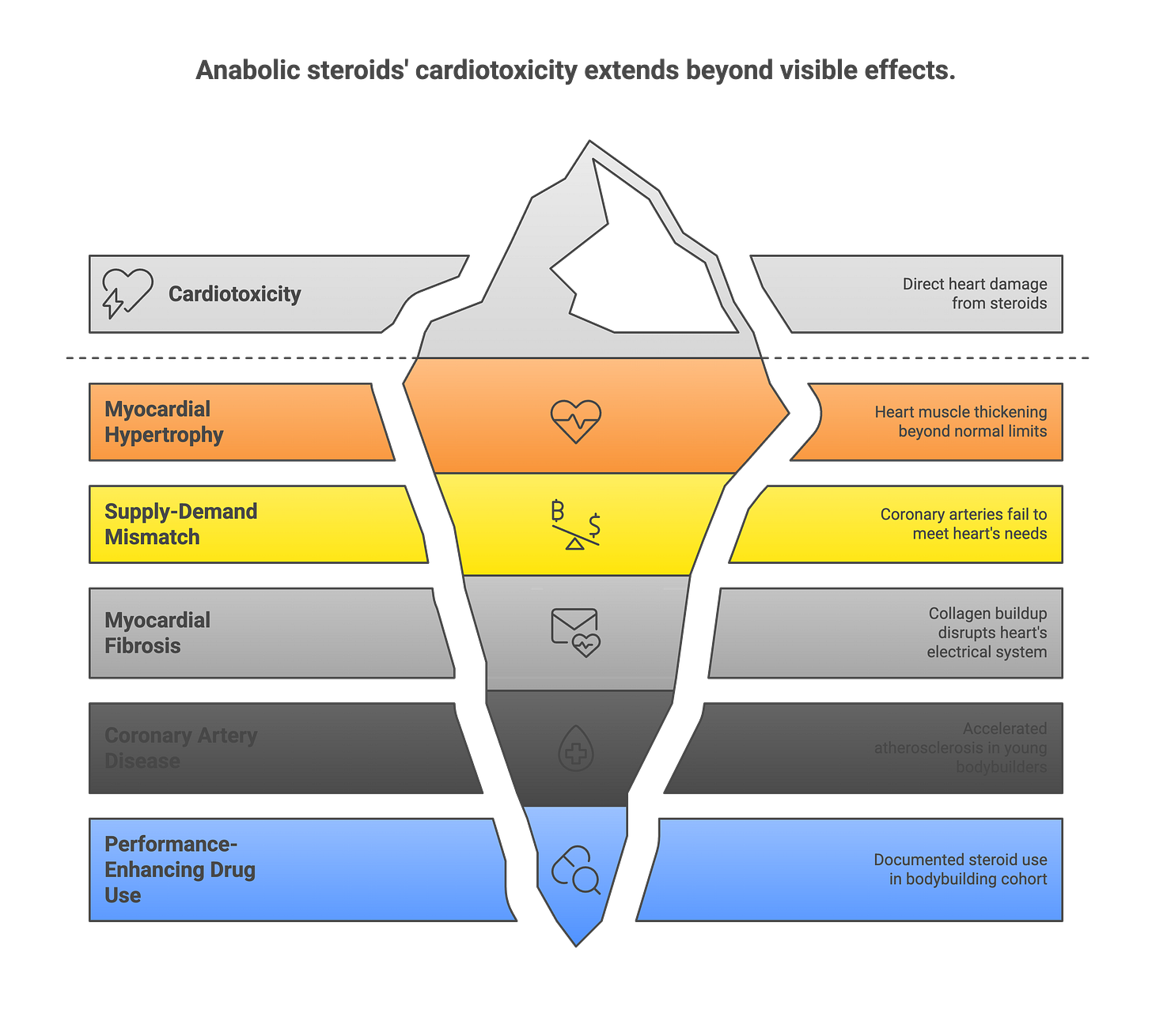
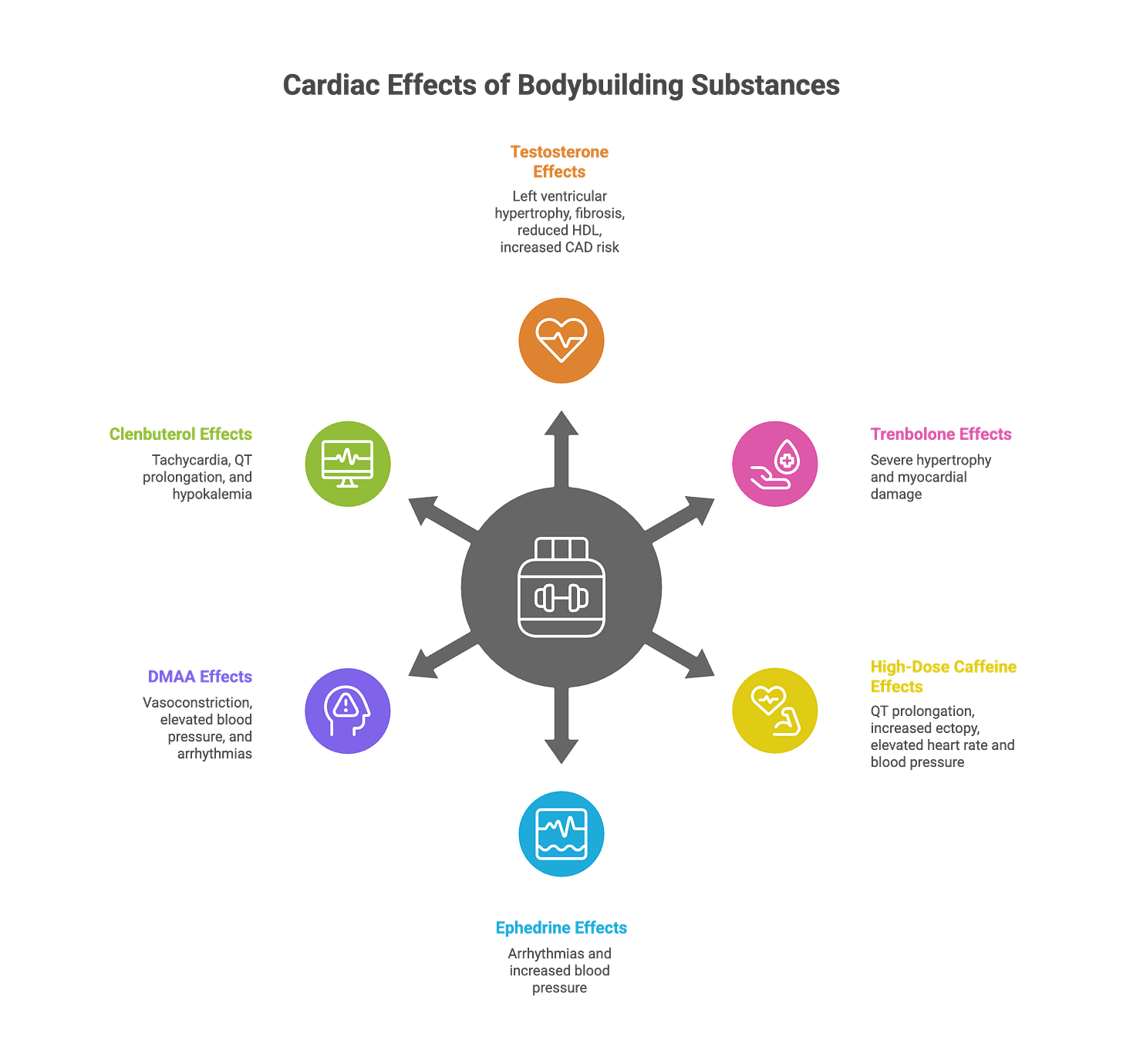
The bodybuilding mortality study’s autopsy data revealed hearts that weighed 73.7% more than normal. This isn’t physiological athlete’s heart. This is pathological hypertrophy—and anabolic-androgenic steroids (AAS) are the primary driver.
Anabolic steroids cause direct cardiotoxicity through multiple mechanisms. They induce left ventricular hypertrophy that exceeds physiological adaptation. The myocardium thickens, but the coronary vasculature doesn’t keep pace—creating a supply-demand mismatch. Collagen deposition increases, leading to myocardial fibrosis. This fibrotic tissue creates the substrate for re-entrant ventricular arrhythmias.
AAS accelerate coronary artery disease. They reduce HDL cholesterol, increase LDL oxidation, and promote endothelial dysfunction. Young bodybuilders in their 30s can develop coronary atherosclerosis resembling that of sedentary 60-year-olds. The autopsy reports from the bodybuilding study documented significant coronary artery disease in athletes decades younger than typical CAD patients.
The toxicology findings tell the story: anabolic steroids were detected in three of five tested cases. At least 16 athletes in the cohort had documented performance-enhancing drug use. The real number is certainly higher—many compounds clear rapidly, and post-mortem testing has limitations.
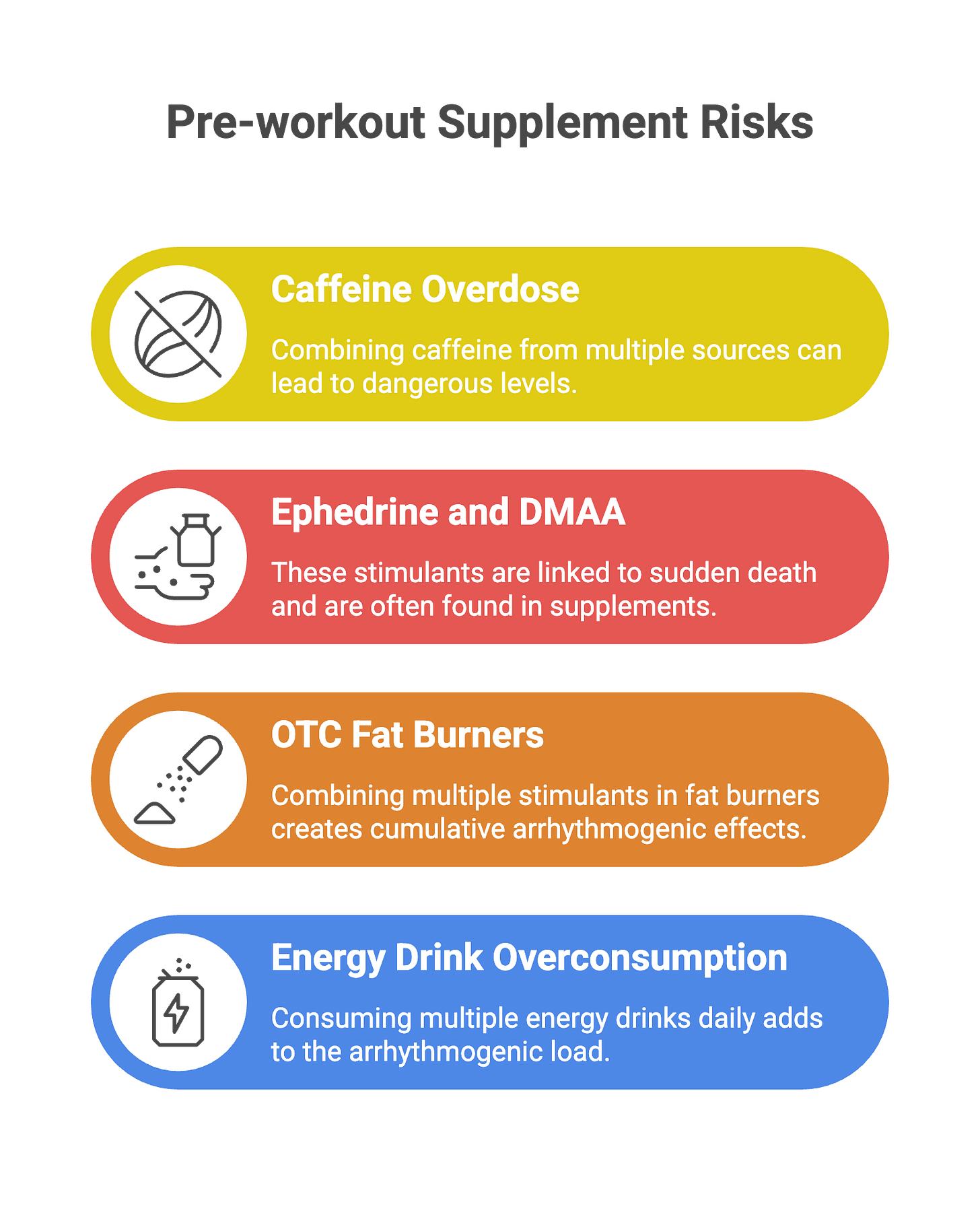
But steroids aren’t the only problem. Pre-workout supplements have become ubiquitous in gym culture, and many contain arrhythmogenic stimulants.
Caffeine in moderate doses (200-400 mg) is relatively safe. The problem is dose-stacking. A pre-workout powder with 300 mg caffeine, combined with an energy drink (160 mg), plus a fat burner (200 mg), delivers 660 mg—enough to prolong the QT interval and increase ectopic beat frequency.
Ephedrine and DMAA (1,3-dimethylamylamine) are more dangerous. Ephedrine increases catecholamine release, raises heart rate and blood pressure, and has been linked to sudden death in young adults. DMAA was banned by the FDA in 2013, but it still appears in supplements sold through online channels. Both compounds synergize with exercise-induced catecholamine surges.
OTC fat burners often combine multiple stimulants—caffeine, synephrine, yohimbine—creating cumulative effects. Add energy drinks to the mix (some bodybuilders consume 3-4 cans daily), and the arrhythmogenic load becomes substantial.
Substance Mechanism Cardiac Effect
The perfect storm occurs when underlying cardiac pathology (HCM, ARVC, ion channelopathy) meets substance-induced remodeling (AAS) and acute sympathomimetic stress (pre-workout stimulants + exertion). A vulnerable heart, a hypertrophied myocardium with fibrotic substrate, a surge of catecholamines—and ventricular fibrillation follows.
So what about the myths flooding your WhatsApp?
4. Debunking the myths
COVID vaccines, whey protein, and ‘overexercising’—what the evidence shows
The viral forwards blame three things: COVID vaccines, whey protein, and “too much exercise.” The data doesn’t support any of these narratives.
COVID vaccines and sudden cardiac death:
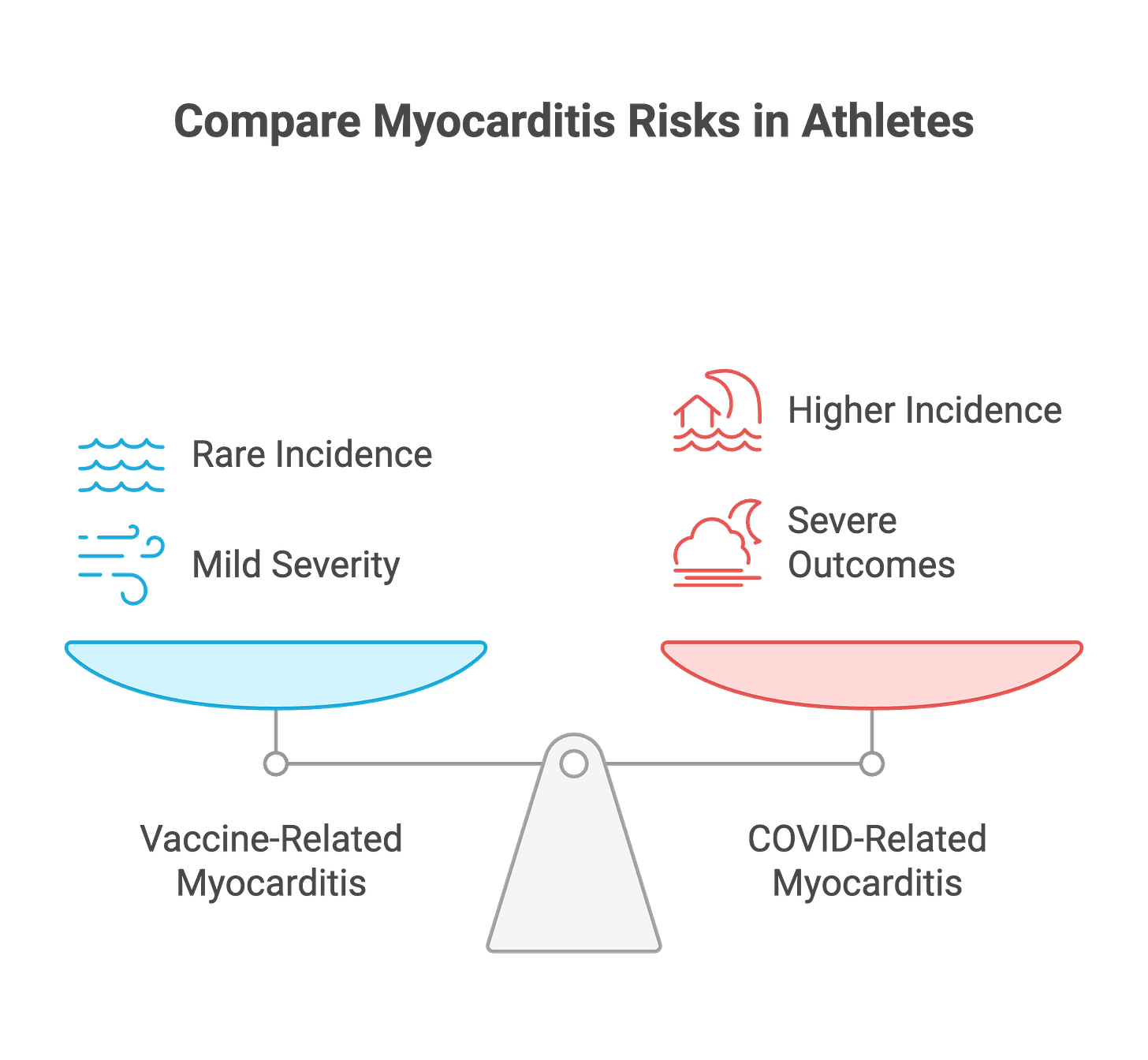
The largest study examining this question tracked 1,229 Team USA athletes competing in the Tokyo 2020 and Beijing 2022 Olympics and Paralympics.[4] All athletes were fully vaccinated by Beijing 2022. The study followed them for over a year post-vaccination, monitoring for cardiac events.
The result: zero sudden cardiac arrests, zero sudden cardiac deaths, zero cases of myocarditis, zero cases of pericarditis.
The study was published in Sports Health in 2023. The conclusion was unequivocal: “Complete absence of vaccine-related cardiac complications in elite athletes.”
Vaccine-related myocarditis does occur, but it’s rare—between 2 and 20 cases per 100,000 vaccinated individuals.[5] When it does occur, it’s typically mild and self-limiting. The risk of myocarditis from COVID infection itself is higher—approximately 40 per million infections. For athletes who develop vaccine-related myocarditis, the recommendation is 3-6 months of exercise abstinence, after which full recovery is typical.
The narrative that COVID vaccines caused the bodybuilder deaths doesn’t fit the timeline or the evidence. If vaccines were the culprit, we’d see a spike in sudden cardiac death across all vaccinated athlete populations, not just bodybuilders with documented anabolic steroid use.
Whey protein and cardiac death:
There is no evidence in the medical literature linking standard whey protein supplementation to sudden cardiac death. Whey protein is a food product—a byproduct of cheese production. Consuming 20-40 grams per day doesn’t cause cardiomyopathy, coronary artery disease, or arrhythmias.
The confusion arises because bodybuilders consume whey protein and anabolic steroids and pre-workout stimulants. The visible supplement (the protein powder) gets blamed, while the invisible ones (the injectable testosterone, the oral DMAA) cause the actual damage.
The bodybuilding mortality study’s toxicology findings identified anabolic steroids, not protein powder. The autopsy data showed pathological cardiac hypertrophy and fibrosis—findings consistent with AAS use, not whey consumption.
“Overexercising”:
The narrative that “too much exercise” killed these athletes is partially correct but misleading. Exercise volume alone doesn’t cause sudden cardiac death in healthy hearts.
The problem is the interaction between exercise, underlying cardiac pathology, and substances.
In athletes with HCM, intense exertion triggers arrhythmias by increasing contractility in an already hypertrophied, disorganized myocardium. In athletes with ARVC, repeated exercise accelerates fibrofatty replacement. In athletes with ion channelopathies, the catecholamine surge during a max lift unmasks electrical instability.
Add anabolic steroids—which create pathological hypertrophy and fibrosis—and you’ve converted a normal heart into a vulnerable one. Add pre-workout stimulants, and you’ve amplified the arrhythmogenic trigger.
A 2024 study in the Journal of Clinical Medicine examined long-distance runners and found that excessive training volumes can lead to exercise-induced hypertension, reduced endothelial function, and increased arterial stiffness.[6] This increases the risk of atrial fibrillation and ventricular arrhythmias—but these effects are seen in extreme endurance training over years, not in typical gym-goers.
The bodybuilders who died weren’t just “exercising too much.” They were exercising intensely with underlying cardiac pathology (HCM, ARVC) and substance-induced remodeling (AAS) and acute sympathomimetic stress (pre-workout stimulants). That’s not overexercising—that’s a multi-hit model for sudden death.
Given this evidence, what can be done?
5. Screening and prevention
Why your cardiologist can’t predict sudden death (and what that means for you)
Varinder Singh Ghuman’s death had no warning signs. No prior symptoms. No collapse during a previous workout. Just sudden death.
This is the screening problem.
HCM can be silent until it isn’t. Up to 30% of HCM cases present with sudden death as the first symptom. The disease can be missed on routine physical examination. Even electrocardiograms (ECGs) have limitations—they detect HCM in only 50-70% of cases. The gold standard is cardiac MRI, but obtaining an MRI for every gym-goer isn’t feasible or cost-effective.
ARVC is even harder to detect in early stages. The fibrofatty replacement begins in the right ventricle, which is difficult to image with echocardiography. Cardiac MRI is more sensitive, but again, population-wide screening isn’t practical.
Ion channelopathies can be genetically silent. A resting ECG might be normal. The abnormalities only appear during exercise stress testing or with provocation testing (epinephrine infusion for Long QT, ajmaline challenge for Brugada). Genetic testing can identify mutations, but not everyone with a pathogenic variant develops clinical disease.
Coronary anomalies require imaging—typically CT angiography or cardiac MRI. They won’t be detected on ECG or echocardiography.
So how do we screen?
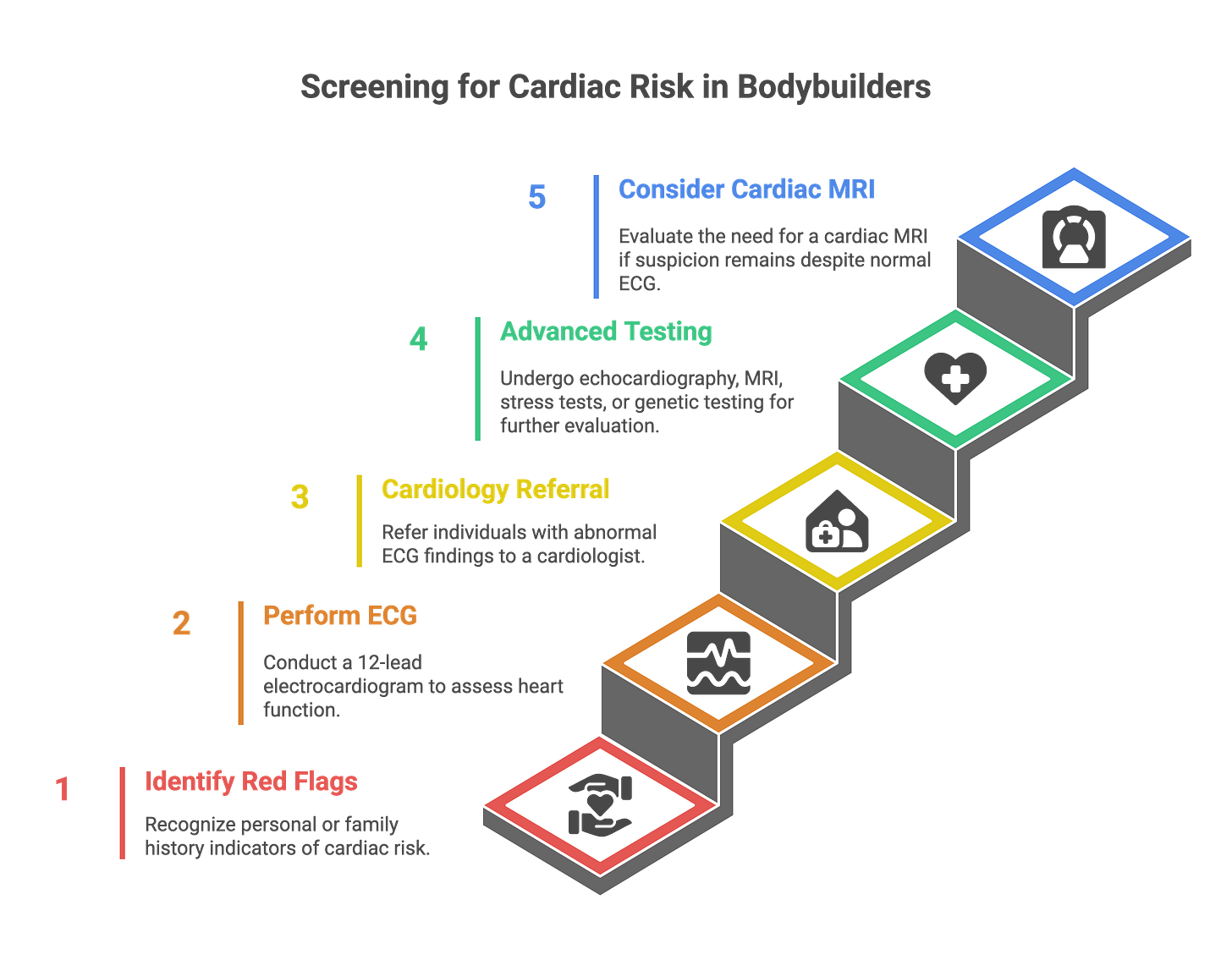
Pre-participation screening for competitive athletes typically includes:
Detailed personal and family history
Physical examination
12-lead ECG
The family history is critical. Sudden death in a first-degree relative under age 50 is a red flag. Unexplained syncope during exertion, chest pain, or palpitations warrant further investigation.
The ECG can identify some cases of HCM (voltage criteria for LV hypertrophy, deep T-wave inversions), ARVC (epsilon waves, T-wave inversions in right precordial leads), and Long QT (prolonged QTc interval). But it misses cases too.
Screening considerations:
Know your family history. If sudden cardiac death runs in your family, you need screening before intense training.
Audit your supplement cabinet. Anabolic steroids cause pathological cardiac remodeling. Pre-workout stimulants containing DMAA, ephedrine, or excessive caffeine increase arrhythmia risk. If you’re using AAS, you need cardiac monitoring—baseline ECG, echocardiogram, and periodic follow-up.
Pay attention to symptoms. Chest pain, pressure, or tightness during exertion isn’t normal. Palpitations that persist after exercise aren’t normal. Syncope or near-syncope during lifting isn’t normal. These symptoms demand evaluation—ECG, echocardiogram, possibly cardiac MRI or stress testing.
Age matters. If you’re over 35 and pushing high-intensity training, CAD screening (stress testing, calcium scoring, or CT angiography) becomes relevant. The bodybuilding mortality data showed that older athletes died from a mix of cardiomyopathies and coronary disease.
Push back on trainers who dismiss symptoms. “No pain, no gain” doesn’t apply to cardiac symptoms. If your chest hurts during a set, stop. If you feel lightheaded or your heart races irregularly, stop. These are warning signs, not weakness.
The collapse-to-defibrillation window determines survival. Data from the sideline management study shows that immediate defibrillation (within 3-5 minutes) is the greatest predictor of survival in sudden cardiac arrest.[3] Gyms should have automated external defibrillators (AEDs) accessible, and staff should be trained in CPR and AED use.
But prevention is better than emergency response. Screening identifies at-risk individuals before they collapse.
Conclusion
The deaths of Varinder Singh Ghuman and five other bodybuilders within a year weren’t caused by COVID vaccines, whey protein, or “too much exercise.” The evidence points to the intersection of three factors: underlying cardiac pathology (HCM, ARVC, ion channelopathies, CAD), substance-induced remodeling (anabolic steroids creating pathological hypertrophy and fibrosis), and acute triggers (intense exertion combined with pre-workout stimulants).
The bodybuilding mortality study’s findings are stark—sudden cardiac death incidence of 32.83 per 100,000 athlete-years in competing bodybuilders, with professional athletes facing a 5.23-fold higher risk. The autopsy data revealed hearts that weighed 73.7% more than normal, with severe hypertrophy and fibrosis creating the substrate for fatal arrhythmias.
The question isn’t whether you should exercise. The question is whether you’re screening for silent cardiac conditions that can turn a workout into a fatal event.
If you found this useful, please share it.
Evidence over opinions. Clarity over noise. That’s Signal Over Noise.
References
Mantel Ä, Holmberg M, Andersson DC, et al. Mortality among male bodybuilders: a cohort study. Eur Heart J. 2025;46(3):241-250. doi:10.1093/eurheartj/ehaf285
Maron BJ, Desai MY, Nishimura RA, et al. Diagnosis and evaluation of hypertrophic cardiomyopathy: JACC State-of-the-Art Review. J Am Coll Cardiol. 2024;83(5):1-24. doi:10.1016/j.jacc.2023.10.032
Yeo TJ, Sharma S. Sideline management of sudden cardiac arrest in athletes. Video J Sports Med. 2025;5:26350254251329104. doi:10.1177/26350254251329104
Daniels CJ, Rajpal S, Greenshields JT, et al. Cardiovascular outcomes following COVID-19 vaccination among elite athletes: 1-year follow-up analysis of Team USA participants. Sports Health. 2023;15(6):795-801. doi:10.1177/19417381231208677
Bozkurt B, Kamat I, Hotez PJ. Myocarditis with COVID-19 mRNA vaccines. Curr Cardiol Rep. 2022;24(10):1383-1391. doi:10.1007/s11886-022-01801-6
Małek ŁA, Barczuk-Falęcka M, Werys K, et al. Exercise-induced blood pressure in recreational long-distance runners and its relationship to arterial stiffness and cardiac biomarkers. J Clin Med. 2024;13(8):2184. doi:10.3390/jcm13082184






Sir,does this mean if someone wants to start gymming (for regular exercise not for professional competition) , a cardiac evaluation should be performed ?
This is very good stuff doc! Thanks for penning this down.