Statins and New-Onset Diabetes: What the Evidence Actually Shows
The Inconvenient Truth We Can’t Ignore—And Why It Still Doesn’t Change What You Should Do
Statins have earned their reputation the hard way - reliably lowering LDL cholesterol, preventing heart attacks, and saving lives across trial after trial. Their track record speaks for itself : few drug classes in medicine can claim such consistent, reproducible evidence across dozens of trials and hundreds of thousands of patients. Yet a persistent finding has emerged from this same body of evidence, one that makes patients and physicians alike uncomfortable: statins modestly increase the risk of developing type 2 diabetes.
This is not a fringe concern. It’s not an artifact of cherry-picked data. And it’s not something we should dismiss with a wave of the hand. The question isn’t whether this effect exists—it does—but rather what it means, why it happens, and how we should incorporate it into clinical decision-making.
The Data That Changed the Conversation
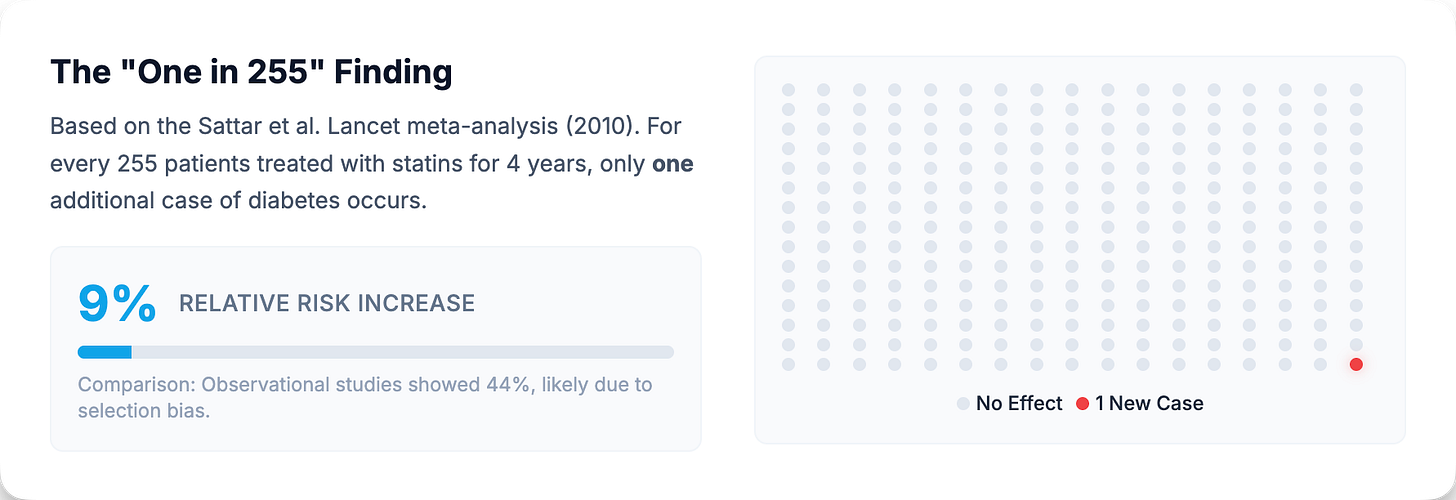
The alarm bells first rang clearly in 2010 when Sattar and colleagues published a landmark meta-analysis in The Lancet, pooling individual patient data from 13 major statin trials involving over 91,000 participants (1). The finding: statin therapy was associated with a 9% increased risk of developing diabetes (odds ratio 1.09; 95% CI 1.02–1.17). In absolute terms, treating 255 patients with a statin for four years resulted in one additional case of diabetes.
That 9% figure has proven remarkably durable. Subsequent analyses have either confirmed it or, in some cases, suggested the risk may appear even higher in real-world populations. A 2017 meta-analysis of 20 observational studies found a 44% increased risk of new-onset diabetes among statin users compared with non-users (relative risk 1.44; 95% CI 1.31–1.58) (2).
However, this observational estimate should be interpreted cautiously. The dramatic discrepancy between the 9% RCT estimate and the 44% observational figure likely reflects confounding by indication—the very factors that lead physicians to prescribe statins (obesity, metabolic syndrome, elevated cardiovascular risk) also independently predispose patients to diabetes. Observational studies also suffer from surveillance bias: patients on statins receive more frequent blood tests, so diabetes gets detected earlier. The RCT estimate of 9% is almost certainly closer to the true causal effect, while the larger observational signal reflects the reality that statin-treated populations already carry elevated metabolic risk.
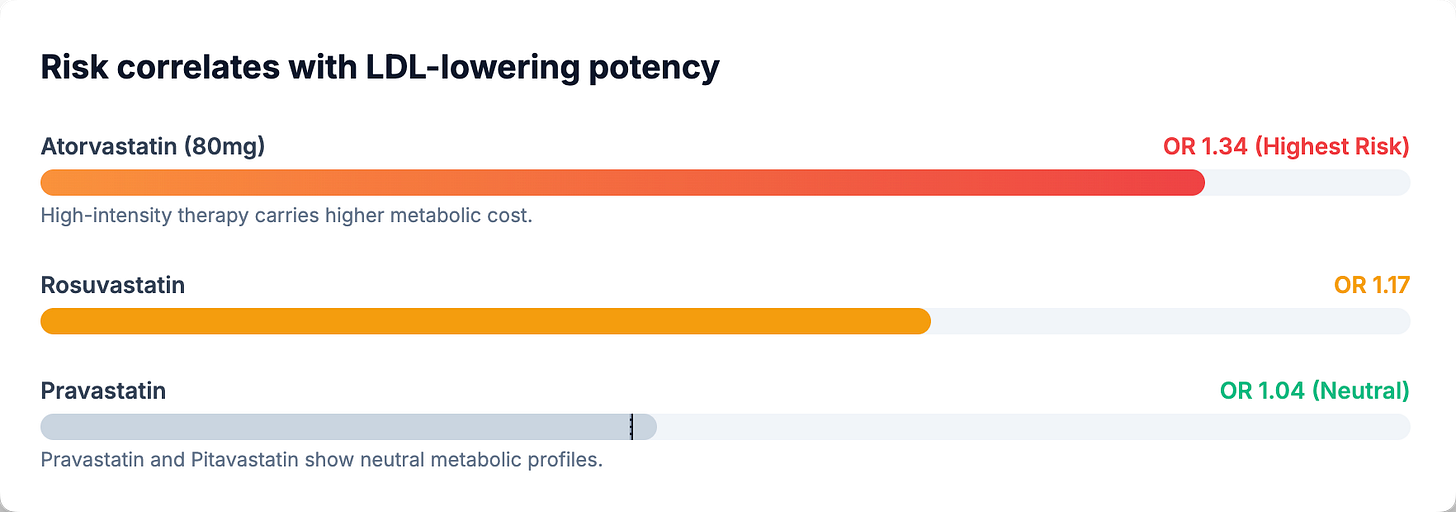
What’s more, the effect appears dose-dependent. A network meta-analysis of 29 trials examining individual statins found that high-dose atorvastatin (80 mg) carried the highest diabetes risk (OR 1.34; 95% CI 1.14–1.57), followed by rosuvastatin (OR 1.17; 95% CI 1.02–1.35) (3). Pravastatin appeared genuinely neutral (OR 1.04; 95% CI 0.93–1.16). Data on pitavastatin suggest possible neutrality, though estimates remain imprecise due to limited trial data (OR 0.74; 95% CI 0.31–1.77—a confidence interval spanning nearly six-fold). The overall pattern is consistent with what we’d expect from a mechanism tied to LDL-lowering potency—the more aggressively you lower LDL, the greater the metabolic perturbation.
Why This Happens: Following the Biology
For years, researchers debated whether the diabetes risk was an on-target effect of statins—directly related to how they work—or an off-target side effect unrelated to cholesterol lowering. Mendelian randomization studies have provided strong evidence favoring the on-target hypothesis, though the question isn’t entirely closed.
When researchers examined genetic variants in the HMGCR gene—which encodes the very enzyme that statins inhibit—they found that people with naturally lower HMGCR activity (and therefore lower LDL cholesterol from birth) also had higher glucose levels, higher body weight, and increased diabetes risk (4). This genetic evidence suggests the diabetes signal isn’t an accident or an impurity in the drug—it appears to be linked to the mechanism itself. That said, Mendelian randomization has its own limitations, including potential horizontal pleiotropy (where genetic variants affect outcomes through multiple pathways), and findings from lifelong genetic exposure may not perfectly predict the effects of pharmacological intervention started in middle age.
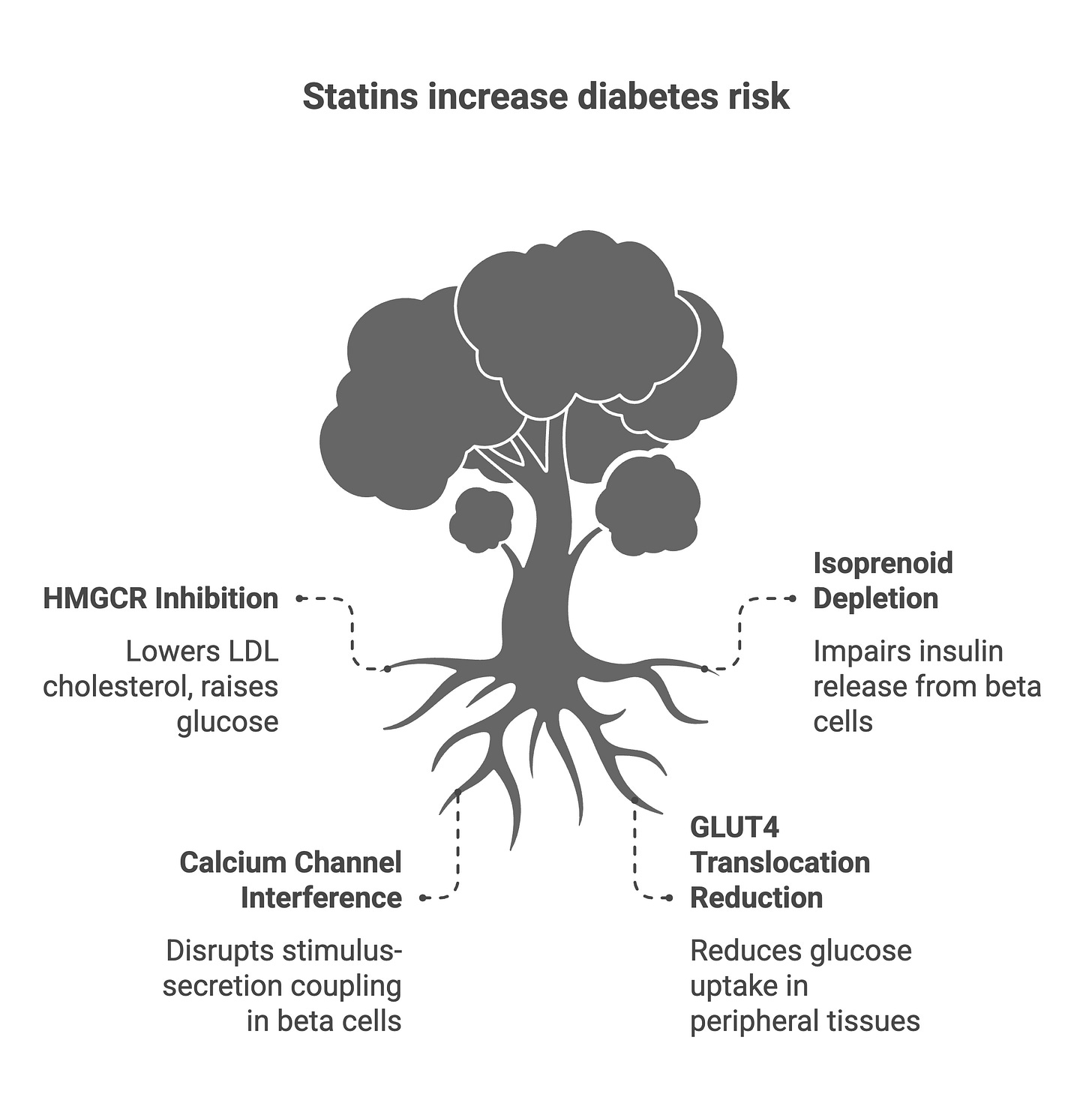
The biological pathways are becoming clearer nonetheless. Statins appear to impair glucose homeostasis through at least two distinct routes, both stemming from inhibition of the mevalonate pathway—the same pathway that produces cholesterol.
First, there’s the effect on insulin secretion. Pancreatic beta cells require certain downstream products of the mevalonate pathway—particularly isoprenoids like geranylgeranyl pyrophosphate—for normal insulin release. When statins block mevalonate synthesis, these isoprenoids become depleted, and beta-cell function suffers (5). There’s also evidence that statins interfere with calcium channel signaling in beta cells, which is essential for stimulus-secretion coupling.
Second, statins impair insulin sensitivity in peripheral tissues. Reduced translocation of glucose transporter 4 (GLUT4) to the cell membrane—the molecular step that allows muscle and fat cells to take up glucose—has been demonstrated in statin-treated cells. The net result: higher circulating glucose and greater insulin demand on an already stressed pancreas (6).
A comprehensive 2023 review in Frontiers in Endocrinology synthesized this evidence, describing a two-pronged mechanism: reduced insulin secretion combined with increased insulin resistance—a combination that nudges susceptible individuals toward diabetes (7).
Who Is Most Vulnerable?
Not everyone faces equal risk. The diabetes signal concentrates among individuals who were already on the path toward metabolic dysfunction. In the JUPITER trial—a large primary prevention study of rosuvastatin—researchers stratified participants by pre-existing diabetes risk factors: metabolic syndrome, impaired fasting glucose, obesity (BMI ≥30), or elevated hemoglobin A1c (8).
Among those with at least one such risk factor, statin therapy was associated with a 28% increase in new-onset diabetes (hazard ratio 1.28; 95% CI 1.07–1.54). Among those with no diabetes risk factors, the increase was essentially absent (HR 0.99; 95% CI 0.45–2.21). The message is clear: if you’re metabolically healthy, statins don’t appear to push you into diabetes. But if you’re already prediabetic, already carrying excess weight, already showing early signs of insulin resistance—statins may accelerate the timeline.
Meta-regression analyses from the original Sattar meta-analysis also identified older age as a risk factor for statin-associated diabetes, though the data on other demographic factors (sex, race) remain less definitive. What we can say with confidence is that the patients at highest metabolic risk at baseline are the ones most likely to develop diabetes on statin therapy—which creates a clinical tension, since these same patients often derive the greatest cardiovascular benefit from treatment.
The Calculus That Matters: Benefits vs. Risks
Here is where clinical judgment enters the picture. We don’t prescribe statins to avoid diabetes—we prescribe them to prevent heart attacks and strokes. The question is whether the cardiovascular benefits outweigh the diabetes hazard.
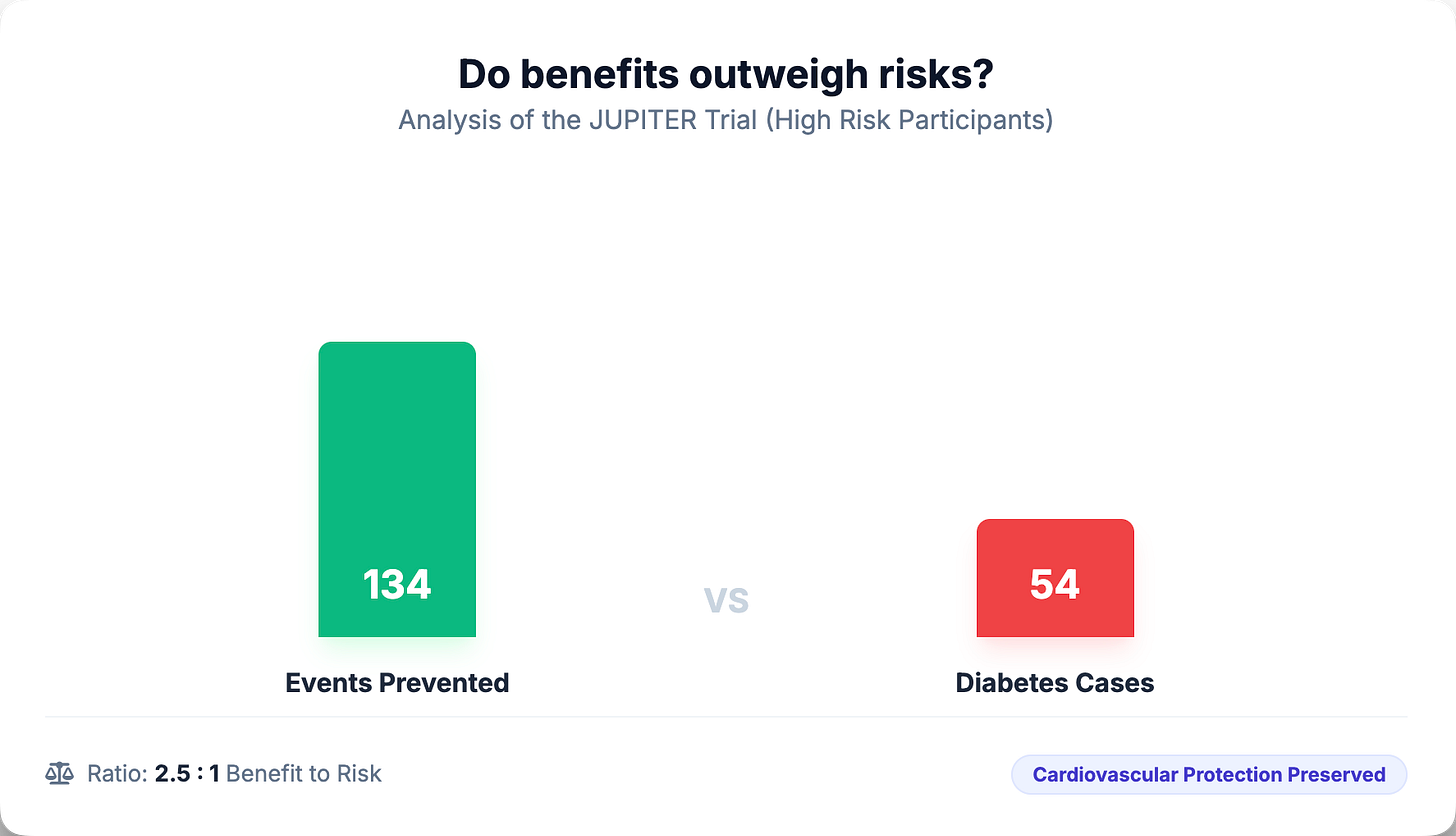
The JUPITER trial provides perhaps the most granular answer. Among participants with diabetes risk factors who took rosuvastatin, 134 vascular events or deaths were prevented for every 54 new diabetes cases diagnosed. That’s roughly a 2.5:1 benefit-to-risk ratio. Among those without diabetes risk factors, 86 vascular events were prevented with no new diabetes cases—essentially all benefit, no metabolic cost.
To put this in perspective: the number needed to treat (NNT) to cause one additional diabetes case over four years is 255. The NNT to prevent one major cardiovascular event in high-risk populations is typically in the range of 20–50. In other words, for every diabetes case induced, statins prevent roughly 5–10 heart attacks, strokes, or cardiovascular deaths. The arithmetic strongly favors treatment.
Moreover, the JUPITER analysis revealed a finding that should reassure patients: even among the 486 participants who developed new diabetes during the trial, the cardiovascular benefit of statin therapy was preserved. Those who developed diabetes on rosuvastatin still saw a 37% relative reduction in vascular events (HR 0.63; 95% CI 0.25–1.60)—a point estimate consistent with the overall trial, though the wide confidence interval reflects the smaller sample size in this subgroup.
What about the nature of the diabetes itself? The JUPITER data showed that statin therapy accelerated the average time to diabetes diagnosis by 5.4 weeks—from 89.7 weeks on placebo to 84.3 weeks on rosuvastatin. One interpretation is that statins unmask latent diabetes rather than creating it de novo, accelerating the diagnosis in people who were already headed toward metabolic dysfunction. However, from the patient’s perspective, earlier onset of a chronic disease remains a meaningful consequence, regardless of whether it was “inevitable.” Both framings have merit: the diabetes is real, but it likely would have developed anyway—just slightly later.
Practical Implications for Patients and Clinicians
None of this should be used as a reason to withhold statins from patients at elevated cardiovascular risk. The data are unambiguous: statins prevent cardiovascular events, and that benefit substantially exceeds the diabetes risk across nearly every population studied.
What this evidence does demand is vigilance. Patients initiating statin therapy—particularly those with metabolic risk factors—should have baseline glucose or A1c measured and periodic reassessment. For most patients, this means an annual metabolic panel. For those with prediabetes, more frequent monitoring may be appropriate.
It also invites a conversation. Patients have a right to understand that statins, like all medications, carry trade-offs. The honest framing is this: Statins modestly increase the likelihood that your body will develop diabetes—most often in people who were already headed that direction. But they substantially decrease your risk of having a heart attack or stroke. For most people at cardiovascular risk, the math strongly favors treatment.
For patients particularly concerned about diabetes risk, lifestyle optimization becomes even more important: weight management, physical activity, dietary improvement. These interventions reduce both cardiovascular risk and diabetes risk simultaneously, and they make the decision to use statins easier by shrinking the potential downside.
Some have asked whether certain statins are “safer” from a metabolic standpoint. The data on pravastatin are reassuring—it appears genuinely neutral on glucose metabolism. Pitavastatin may also be favorable, though the evidence base is thinner and confidence intervals are wide. For patients at particularly high diabetes risk who require statin therapy, these agents deserve consideration, provided equally effective cardiovascular protection can be achieved. However, head-to-head comparative trials powered specifically for diabetes outcomes don’t exist, so switching statins based solely on marginally different metabolic signals remains a judgment call rather than an evidence-based mandate.
The Bottom Line
Statins increase the risk of diabetes. This is not a controversy—it’s an established finding supported by randomized trials, observational studies, and genetic evidence. The effect is modest in absolute terms (roughly 9% relative increase, or one case per 255 patients treated for four years), concentrated among those already at metabolic risk, and appears mechanistically linked to the very pathway through which statins save lives.
What makes this finding tolerable—even acceptable—is the simple arithmetic of benefit and harm. The cardiovascular events prevented far outnumber the diabetes cases induced. And for those who do develop diabetes on statin therapy, the cardiovascular protection persists.
The responsible approach is neither denial nor alarm. It’s transparency: patients deserve to understand both the benefits and the risks of their medications. It’s monitoring: we can catch glucose elevations early and intervene with lifestyle or treatment. And it’s perspective: in the grand calculus of cardiovascular risk, statins remain one of our most valuable tools.
For the millions of patients at elevated cardiovascular risk, the case for statin therapy remains strong. The diabetes signal is real, but it doesn’t change the conclusion—it just reminds us that in medicine, nothing is free.
If you found this analysis valuable, please share it.
Evidence over opinions. Clarity over noise. That’s Signal Over Noise.