Impact of Vaccination and Cardiovascular Health - Implications for Patients with Heart Disease and the Elderly
The Vaccine Debate You Should Actually Be Having
Over the past week, Zoho founder Sridhar Vembu ignited a firestorm by linking childhood vaccines to autism, citing a non-peer-reviewed report that claimed vaccination is “the most significant preventable driver” of autism. India’s medical community responded swiftly, condemning the claims as scientifically baseless and warning that such statements from influential figures risk fueling vaccine hesitancy.
The debate has consumed social media for days. Arguments over vaccine safety, anecdotal accounts of alleged harm, and accusations of credential worship versus evidence-based medicine have dominated discourse.
Meanwhile, a separate evidence base—robust, published in top-tier journals, and endorsed by major cardiology societies—remains largely unimplemented. Influenza vaccination reduces major adverse cardiovascular events by 25-28% in high-risk populations, with cardiovascular mortality reductions of 26-41%. The efficacy is comparable to statins and antihypertensive therapy. Yet vaccination rates among cardiovascular disease patients remain between 27% and 60%.
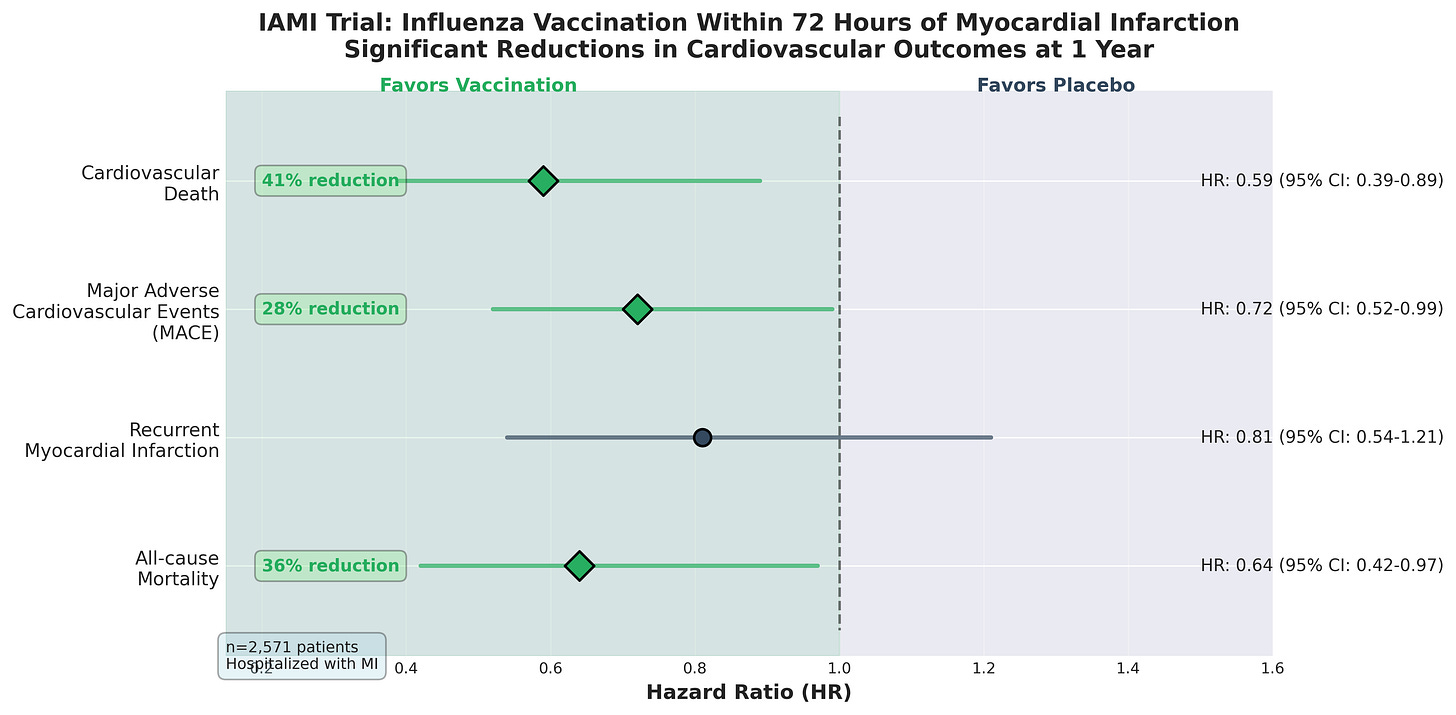
The IAMI trial, published in Circulation in 2022, demonstrated that influenza vaccination within 72 hours of myocardial infarction reduced cardiovascular death by 41%. The mechanism appears to involve immunomodulation during the acute inflammatory period post-infarction, not simply prevention of future respiratory infections.
This is the conversation being drowned out.
The numbers that should have changed everything
In 2022, Swedish researchers tested whether influenza vaccination immediately after myocardial infarction could reduce subsequent cardiovascular events. The IAMI trial enrolled 2,571 patients hospitalized with heart attacks—both ST-elevation and non-ST-elevation MI. Half received the flu vaccine within 72 hours of admission. Half received placebo.
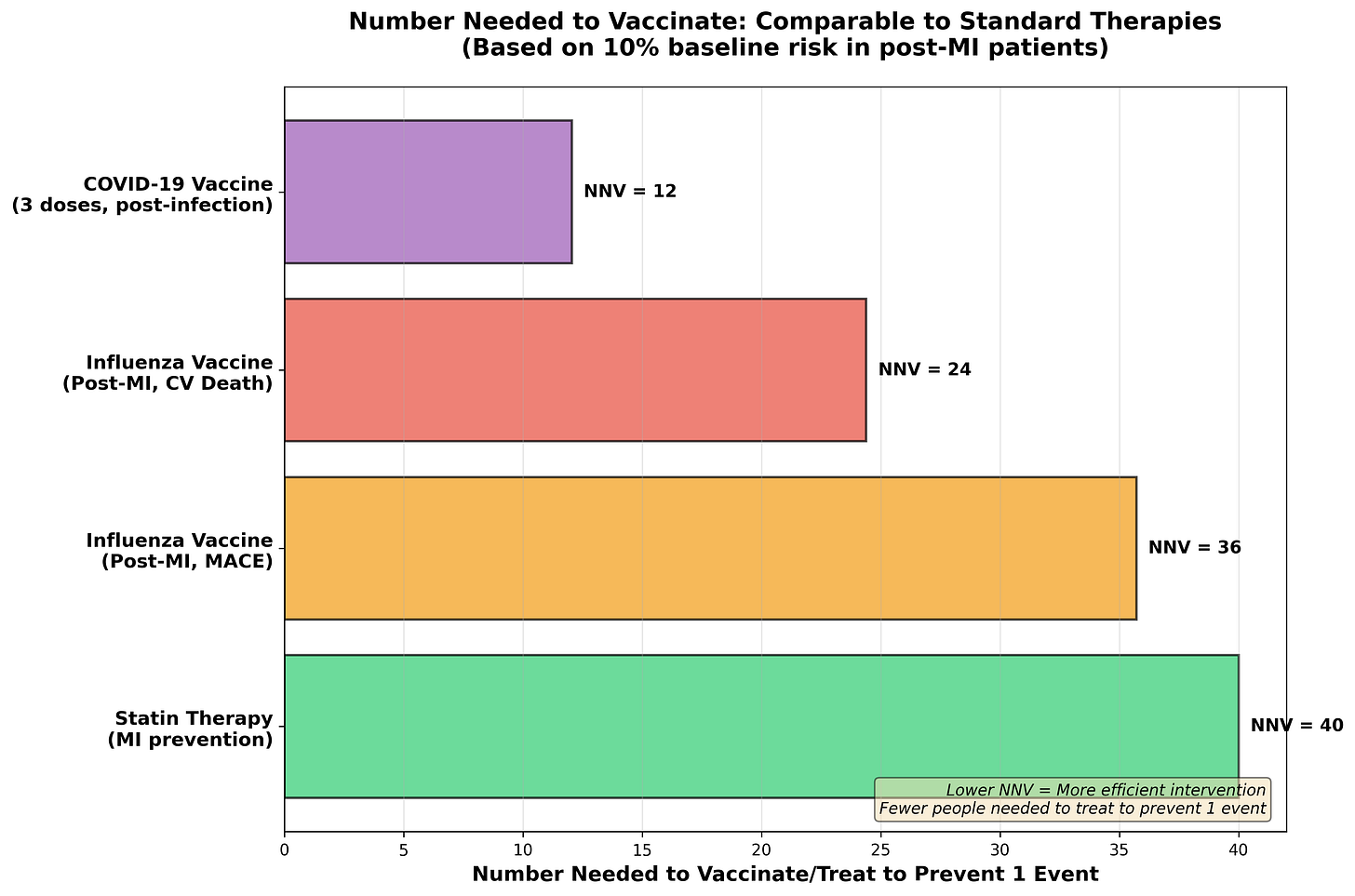
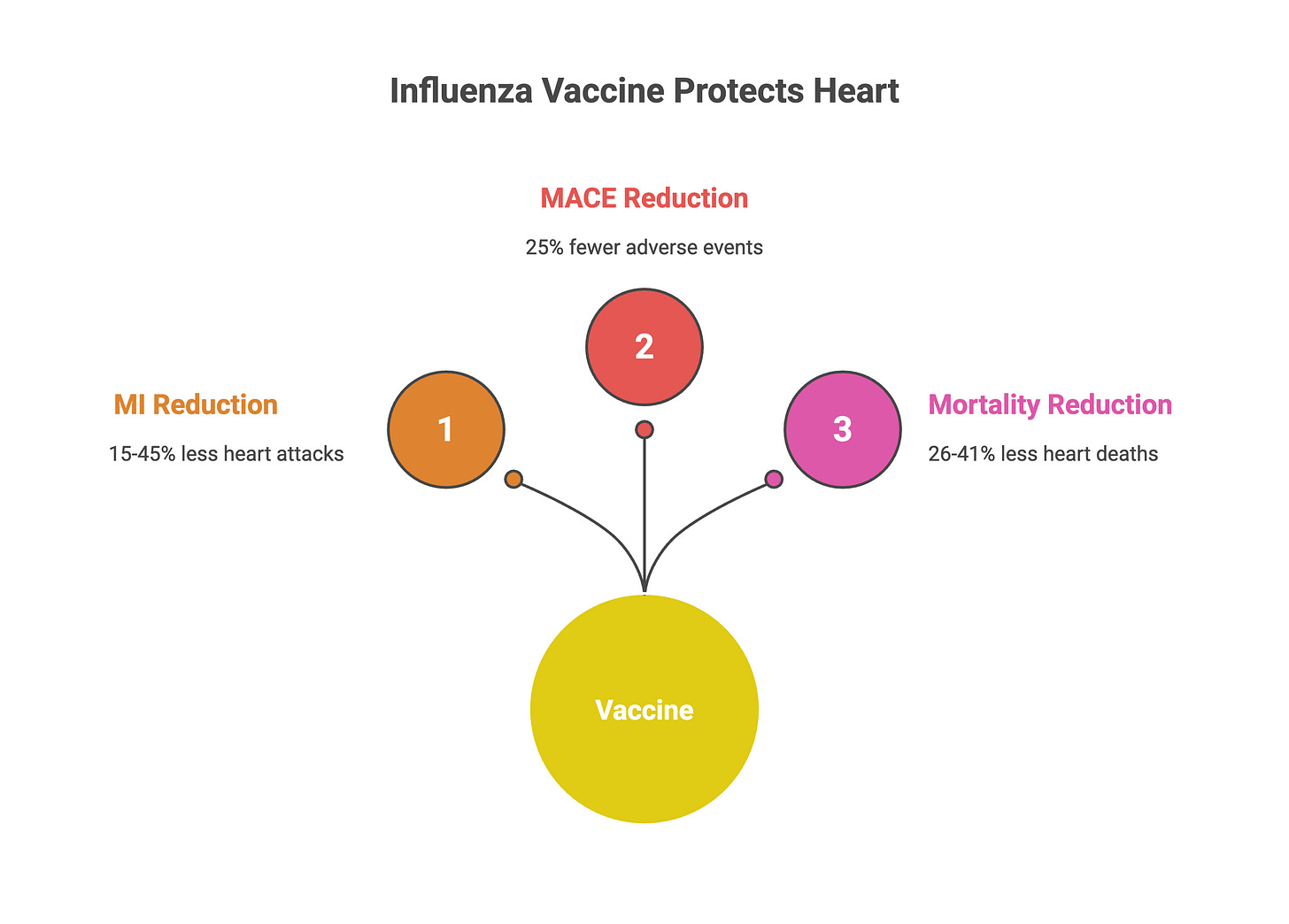
Over the following year, the vaccine group had 28% fewer major cardiovascular events: a composite of death, recurrent heart attack, or stent thrombosis (HR 0.72, 95% CI 0.52–0.99).
The benefit was even more pronounced for cardiovascular death specifically. Vaccinated patients had 41% lower cardiovascular mortality (HR 0.59, 95% CI 0.39–0.89). The effect was consistent whether patients had STEMI or non-STEMI.
This rapid benefit suggests the vaccine isn’t simply preventing seasonal flu infections that might occur later; it’s likely modulating the inflammatory milieu during the acute post-infarction period when atherosclerotic plaques are most unstable and thrombotic risk is highest.
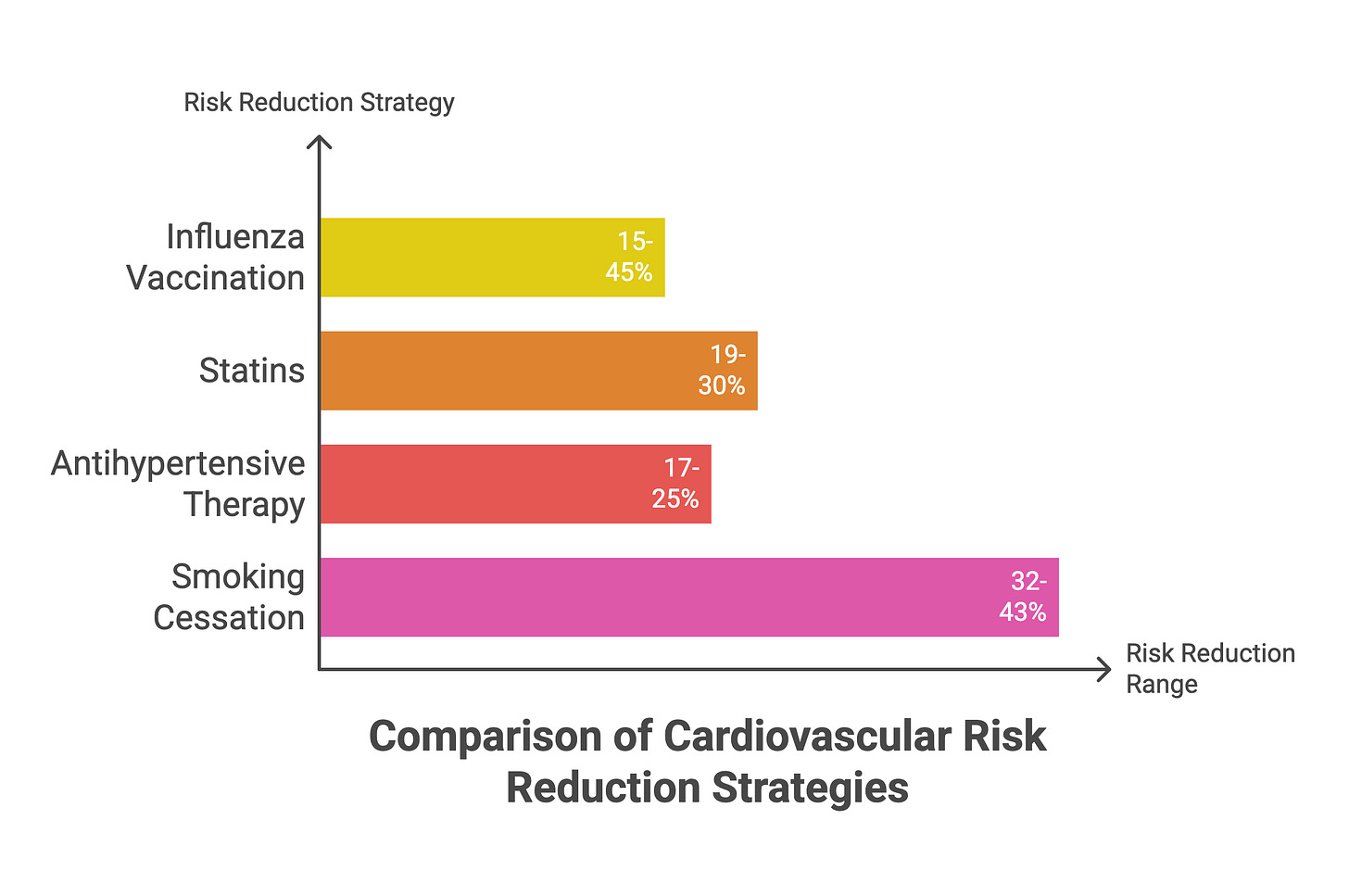
To contextualize the efficacy: influenza vaccination reduces myocardial infarction risk by 15-45% across multiple studies, with cardiovascular mortality reductions ranging from 26-41% in high-risk populations. How does this compare to therapies we already prescribe religiously? Statins reduce MI risk by 19-30%. Antihypertensive therapy reduces risk by 17-25%. Smoking cessation reduces risk by 32-43%. Influenza vaccination sits comfortably within this range—yet it’s treated as optional, an afterthought. Influenza vaccination rates among cardiovascular disease patients hover between 27% and 60% depending on the region and population studied.
To understand why this matters beyond preventing respiratory infections, the biological mechanisms warrant examination.
Vaccines do more than just preventing flu
The cardioprotective effects of vaccination operate through four interconnected mechanisms.
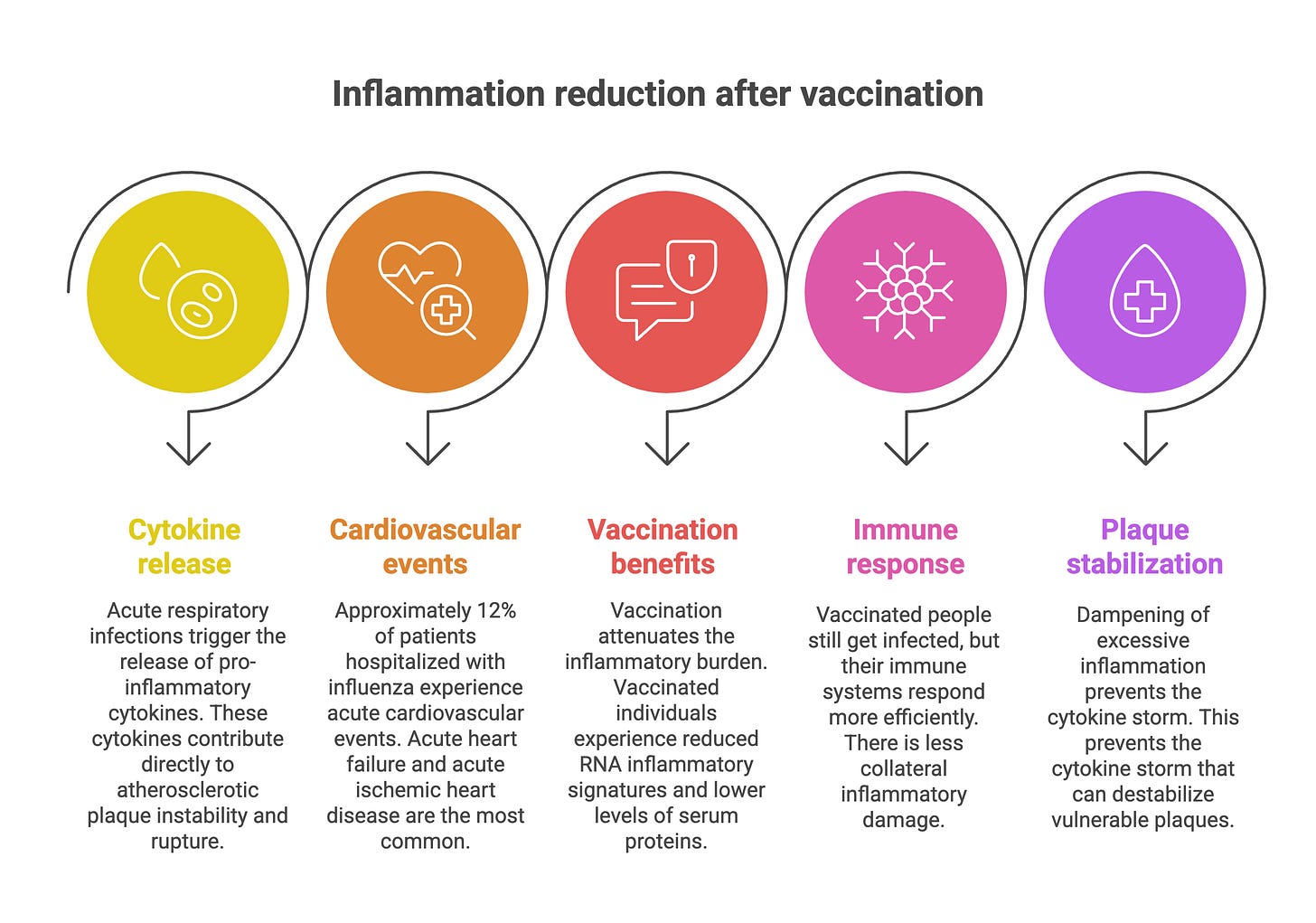
Inflammation reduction. Acute respiratory infections trigger the release of pro-inflammatory cytokines—interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), and IL-1β—that contribute directly to atherosclerotic plaque instability and rupture. Studies show that approximately 12% of patients hospitalized with influenza experience acute cardiovascular events, with acute heart failure (6.2%) and acute ischemic heart disease (5.7%) being the most common. One in four fatal influenza cases has an associated acute cardiovascular event.
Vaccination attenuates this inflammatory burden. Multi-omics analysis of COVID-19 vaccinated individuals who experienced breakthrough infections revealed significantly reduced RNA inflammatory signatures and lower levels of serum proteins associated with disease severity compared to unvaccinated infected individuals. Vaccinated people still got infected, but their immune systems responded more efficiently, with less collateral inflammatory damage. This dampening of excessive inflammation prevents the cytokine storm that can destabilize vulnerable plaques.
Thrombosis prevention. Influenza infection activates the coagulation cascade, increases platelet reactivity, and promotes endothelial dysfunction. The virus can also trigger Type 2 myocardial infarction through increased metabolic demands on myocardial tissue secondary to fever and tachycardia, coupled with potential hypoxemia from respiratory involvement.
Vaccination reduces the incidence and severity of infections, thereby decreasing the thrombotic triggers. Even when breakthrough infections occur in vaccinated individuals, the attenuated inflammatory response translates to lower thrombotic risk.
Trained immunity. This is where the science gets fascinating. Vaccines induce trained immunity—a form of innate immune memory mediated by epigenetic, transcriptional, and functional reprogramming of innate immune cells such as monocytes, macrophages, and natural killer cells. Unlike adaptive immunity (which is antigen-specific), trained immunity enables a more rapid and effective response to subsequent infections, including pathogens unrelated to the original vaccine.
BCG vaccination, for instance, has been shown to provide cross-protection against diverse respiratory pathogens, potentially reducing the cumulative inflammatory burden from multiple infections over time. Research on BCG-induced trained immunity identified metabolic pathways—including the TCA cycle, glutamine metabolism, and linoleic acid metabolism—that correlate with the magnitude of trained immunity responses. Baseline metabolite profiles can predict trained immunity responses months after vaccination.
While most trained immunity research focuses on BCG, influenza and other vaccines likely induce similar epigenetic reprogramming. This could explain heterologous protection: the observation that certain vaccines provide benefits beyond their target pathogen.
Endothelial function. Direct evidence on vaccination’s effects on endothelial function remains limited, but several lines of research suggest potential benefits. Chronic inflammation is a well-established contributor to endothelial dysfunction, a key early step in atherosclerosis progression and a predictor of future cardiovascular events. By attenuating systemic inflammation, vaccination may indirectly improve endothelial function.
COVID-19 vaccination studies demonstrate that vaccines can reduce inflammatory signatures associated with endothelial injury, including downregulation of adhesion molecules and inflammatory markers that contribute to vascular dysfunction. Influenza vaccination has been hypothesized to modulate inflammation important in myocardial infarction and indirectly prevent thrombosis.
These mechanisms apply across multiple vaccines, not just influenza. Four have demonstrated or suggest cardiovascular protective effects.
The vaccines your cardiologist should be recommending (but probably isn’t)
Four vaccines deserve a place in every cardiac patient’s prevention plan. Here’s the evidence for each—and why uptake remains inadequate.
Influenza (annual). This is the most extensively studied vaccine for cardiovascular protection, with decades of research across observational studies, randomized controlled trials, and meta-analyses. The data are unequivocal: influenza vaccination reduces myocardial infarction risk by 15-45%, major adverse cardiovascular events by 25%, and cardiovascular mortality by 26-41% in high-risk populations.
Despite this, only 27-60% of cardiovascular disease patients are vaccinated annually. A systematic review of heart failure patients found influenza vaccination coverage ranging from as low as 2.5% to as high as 97%, with most studies falling below 60%. In elderly patients post-MI, vaccination rates in some regions hover around 58.9%—better than general cardiac populations but still leaving four out of ten patients unprotected.
If a pharmaceutical drug delivered a 28% reduction in major adverse cardiovascular events and a 41% reduction in cardiovascular mortality with minimal side effects, we would prescribe it universally and consider it malpractice not to. Yet we treat the flu vaccine as optional.
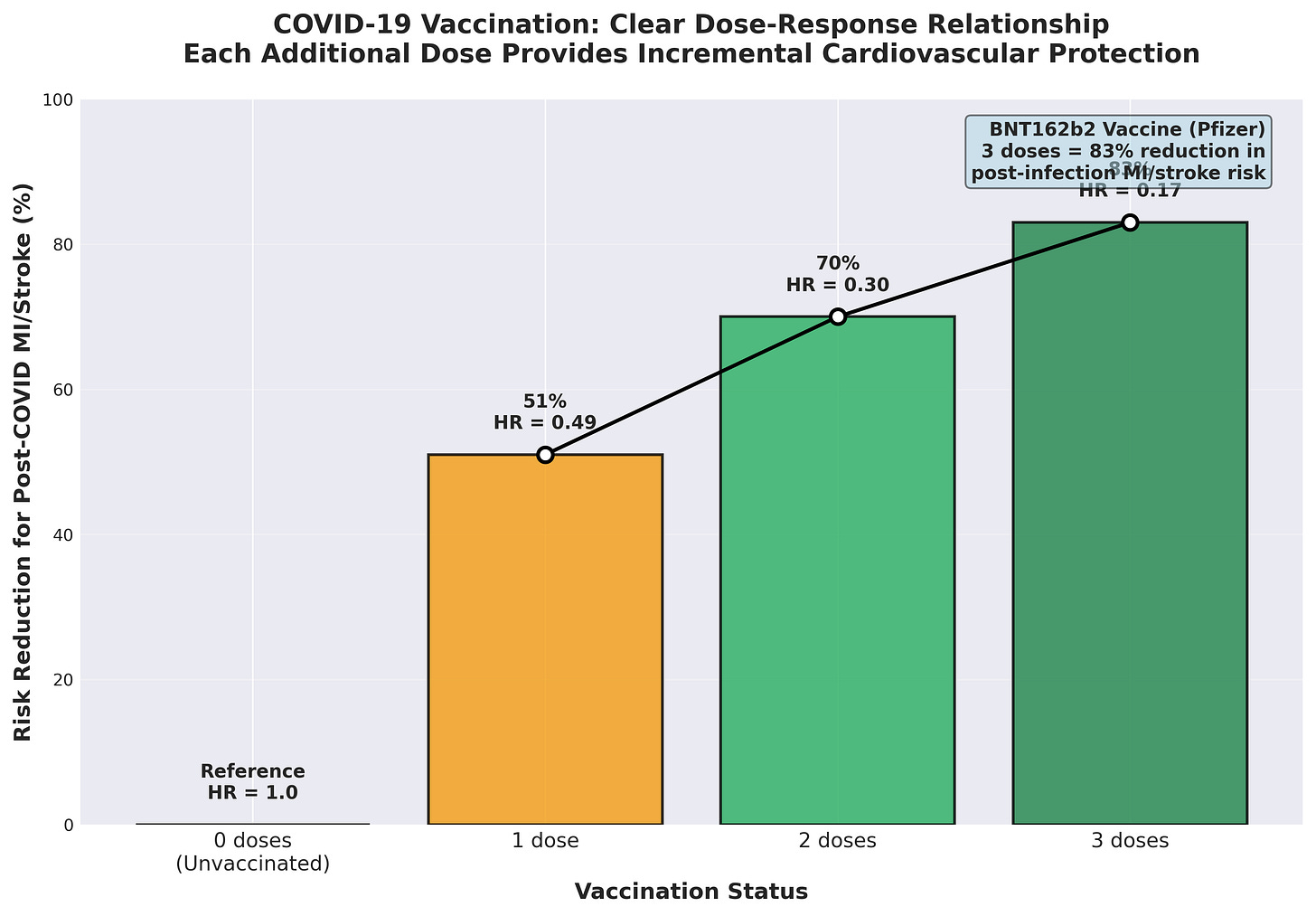
COVID-19 (boosters per CDC guidance). COVID-19 vaccination demonstrates robust cardiovascular protection, particularly in preventing post-infection complications. A large case-control study showed that BNT162b2 or CoronaVac vaccination was associated with lower risk of myocardial infarction or stroke after SARS-CoV-2 infection in patients with cardiovascular disease, with a clear dose-response relationship. For BNT162b2, the risk of post-infection MI or stroke decreased from a hazard ratio of 0.49 (95% CI 0.29–0.84) with one dose to 0.30 (95% CI 0.20–0.44) with two doses and 0.17 (95% CI 0.08–0.34) with three doses. Three doses provide an 83% risk reduction.
A comprehensive meta-analysis demonstrated that COVID-19 vaccination reduced the risk of post-COVID-19 cardiac and thromboembolic outcomes, with meta-analytic subdistribution hazard ratios of 0.22 (95% CI 0.17–0.29) for 0-30 days after SARS-CoV-2 infection. The protective effect was strongest during the acute phase, when cardiovascular complications are most likely.
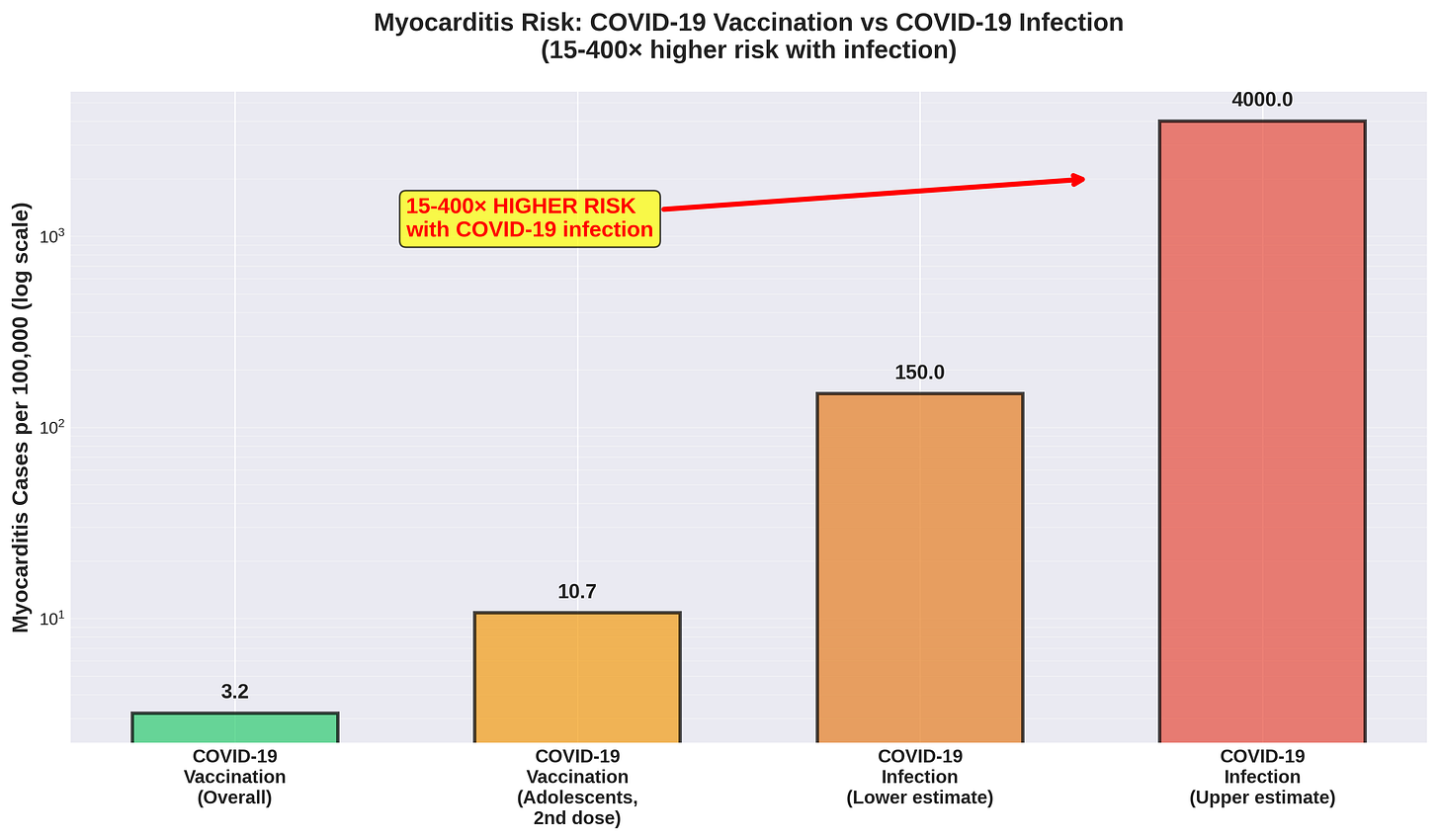
Yes, COVID-19 mRNA vaccines have been linked to myocarditis and pericarditis, particularly in male adolescents and young adults, with incidence rates of approximately 0.0032% overall and up to 0.0107% after the second dose in adolescents. But context matters: the incidence of myocarditis following COVID-19 infection itself is at least 15-fold higher, ranging from 150 to 4,000 cases per 100,000 infections compared to 1-10 cases per 100,000 vaccinations. In older adults—the population at highest cardiovascular risk—myocarditis risk from vaccination is minimal while COVID-19 severity risk is maximal. The risk-benefit calculation strongly favors vaccination
.Coverage data show 68.4% of adults with coronary heart disease received COVID-19 vaccination, with just over half receiving booster doses. This leaves a substantial proportion of high-risk cardiac patients unprotected against a virus that demonstrably triggers cardiovascular events.
Pneumococcal (PCV20 or PCV15 → PPSV23). Pneumococcal vaccination is recommended for patients with heart failure and other cardiovascular conditions based on the rationale that pneumococcal pneumonia is associated with significant increases in hospitalization and mortality rates in CVD patients. Chronic heart disease qualifies patients for pneumococcal vaccination at younger ages than the general population.
The evidence base for cardiovascular-specific outcomes is less extensive than for influenza, but the biological plausibility is sound: invasive pneumococcal disease triggers similar inflammatory cascades to influenza, potentially precipitating acute cardiovascular events. Current guidelines from major cardiology societies recommend pneumococcal vaccination as part of comprehensive cardiovascular risk reduction strategies.
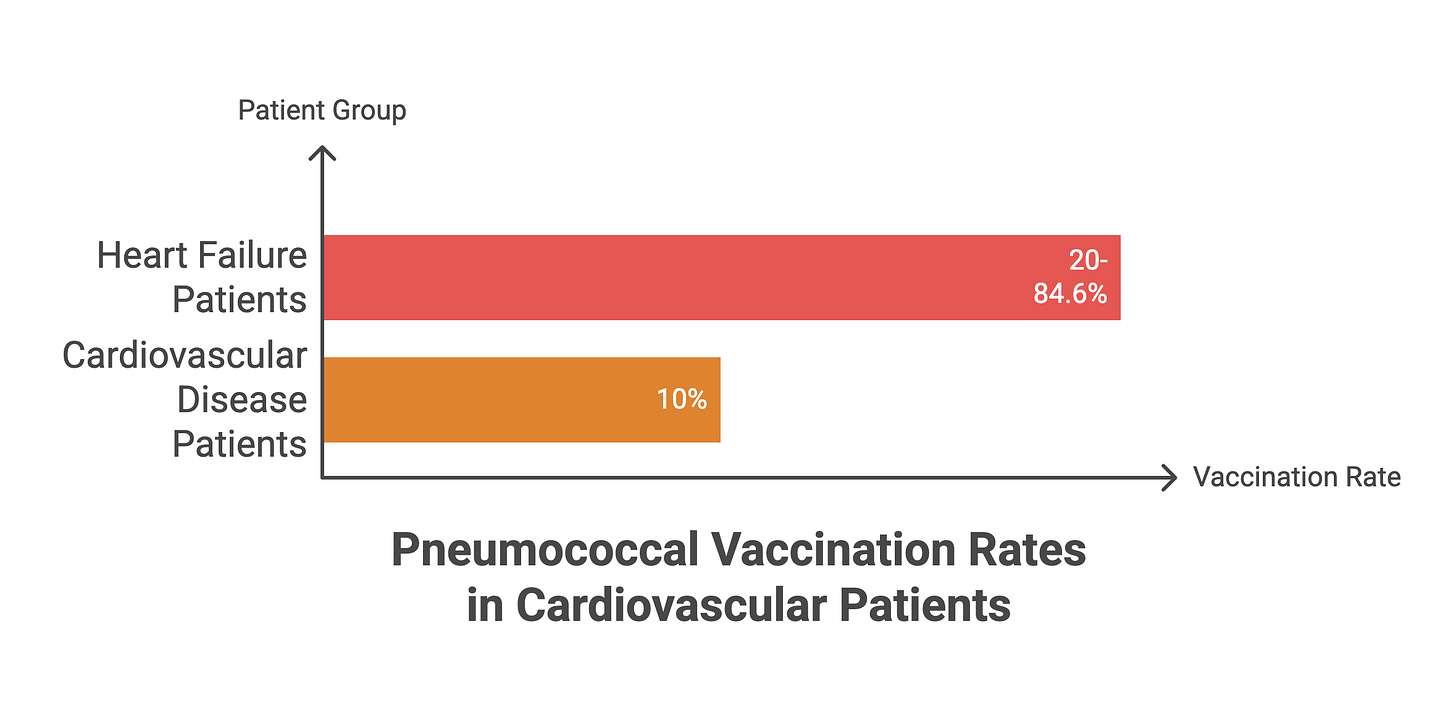
The coverage is abysmal. Pneumococcal vaccination rates among heart failure patients range from 20% to 84.6% across studies, with many falling below 30%. In one study of cardiovascular disease patients, only 10% had received pneumococcal vaccination. For a vaccine with clear guideline support and minimal risk, this uptake represents a massive failure of preventive care.
RSV (one-time, adults ≥60 with risk factors or ≥75). Respiratory syncytial virus vaccines are the newest addition to the cardiovascular prevention arsenal. RSVPreF3 OA, an adjuvanted RSV prefusion F protein vaccine, was recently approved for adults aged 60 and older following successful Phase 3 trials. The DAN-RSV trial specifically examined cardiovascular outcomes in adults ≥60 years.
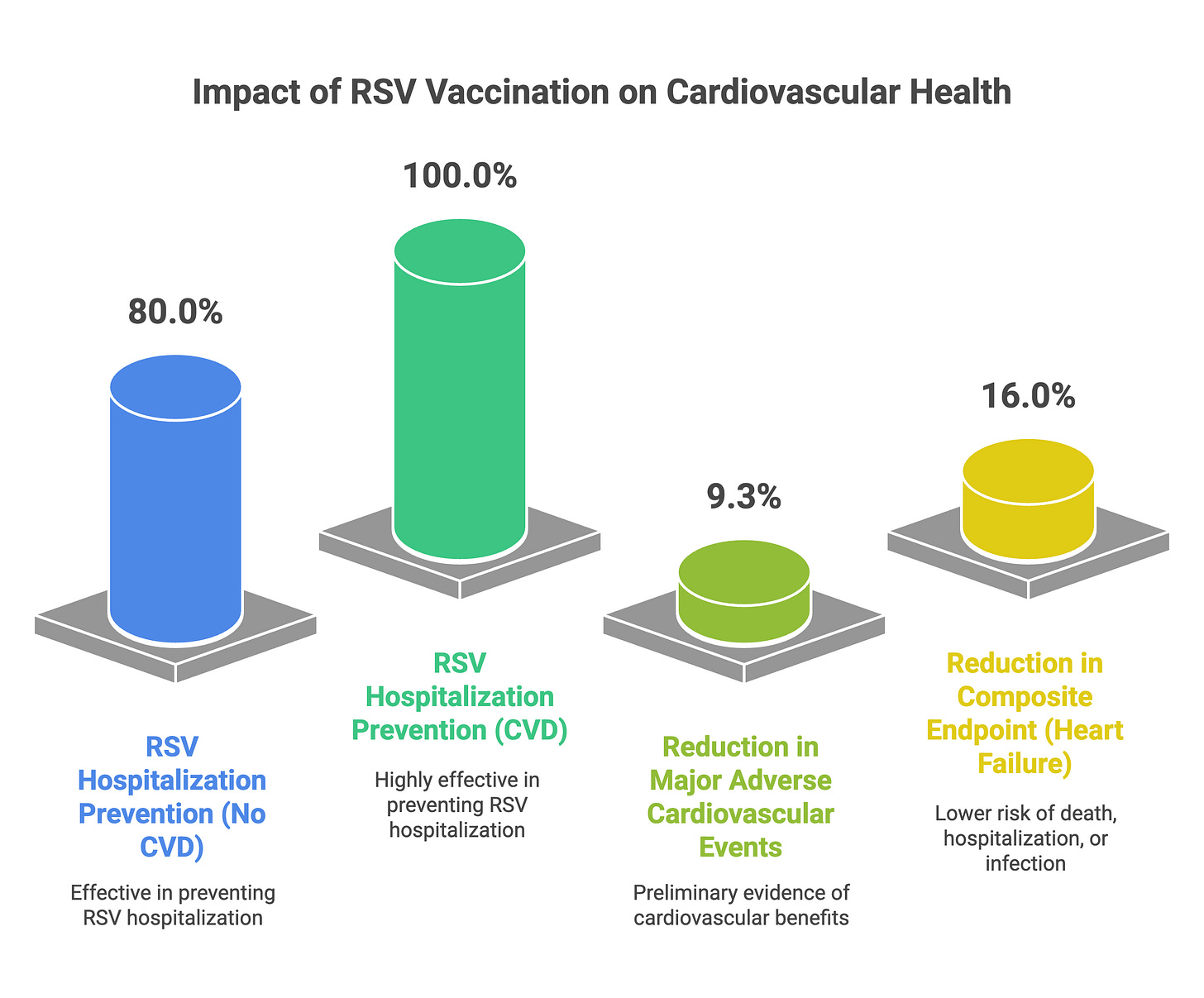
Vaccine effectiveness for preventing RSV-related respiratory tract disease hospitalization was 80.0% (95% CI 29.3–96.3) in participants without pre-existing atherosclerotic cardiovascular disease and 100.0% in those with pre-existing cardiovascular disease (though with wide confidence intervals). Notably, the vaccine demonstrated a 9.3% reduction in major adverse cardiovascular events, though this did not reach statistical significance. This is preliminary but intriguing evidence that RSV vaccination may provide cardiovascular benefits beyond infection prevention.
A randomized trial of simultaneous influenza and RSV vaccination in high-risk heart failure patients demonstrated that the primary composite endpoint of all-cause death, heart failure hospitalization, or clinical signs of infection occurred in 59% of vaccinated patients versus 75% in the standard of care group (HR 0.66, 95% CI 0.48–0.92).
RSV vaccination is currently recommended as a one-time dose (not annual) for adults ≥75 years and for adults 60-74 years with high-risk conditions including chronic heart disease. Uptake data are still emerging, but initial implementation suggests low coverage requiring aggressive public health campaigns.
The pattern is clear: strong evidence, guideline support, low implementation. The question is why.
Why cardiologists aren’t talking about this—and how statements like Vembu’s make it worse
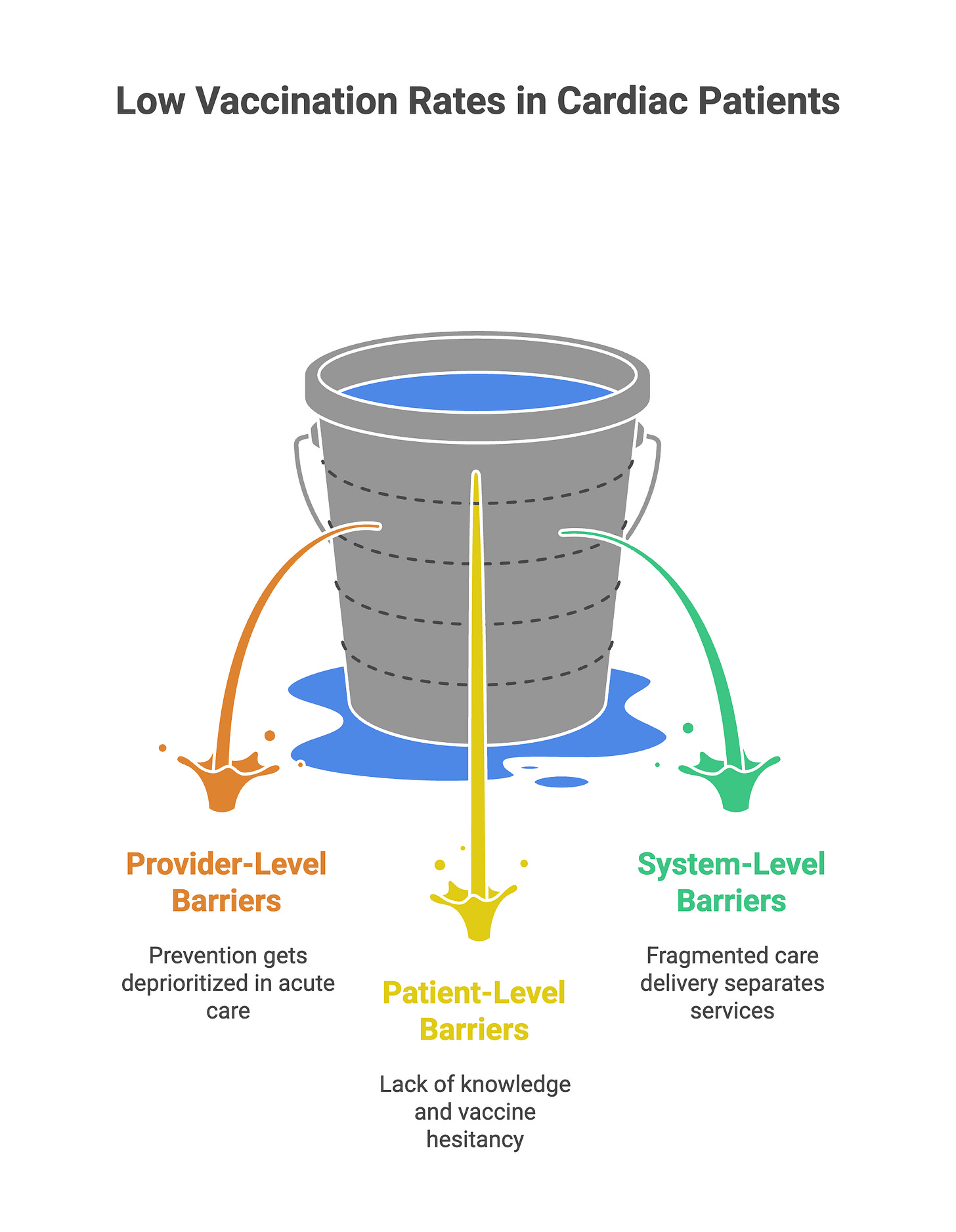
If the evidence for vaccination as cardiovascular prevention is this compelling, why do uptake rates remain so low? The barriers operate at multiple levels—and recent controversies amplify them.
Physician-level barriers. Prevention gets deprioritized in acute care settings. When managing a post-MI patient, the immediate focus is dual antiplatelet therapy, beta-blockers, ACE inhibitors, statins, and cardiac rehabilitation referral. Vaccination doesn’t make the discharge checklist, even though the IAMI trial demonstrates it should. Many cardiologists may not be fully aware of the cardiovascular-specific benefits—the IAMI trial is only from 2022, and most cardiology training programs don’t emphasize vaccines as cardioprotection. A survey found that only 61.6% of cardiac patients received vaccination information from their physicians.
Patient-level barriers. Patients often view vaccines as infection prevention, not heart attack prevention, so they don’t prioritize them. Vaccine hesitancy driven by safety concerns and misinformation disseminated through social media adversely affects uptake. For COVID-19 vaccines specifically, concerns about myocarditis—despite the rare absolute risk—contributed to hesitancy even among high-risk cardiac populations.
This is where controversies like Sridhar Vembu’s become directly harmful. When influential figures amplify discredited vaccine-autism links, they erode trust in vaccination broadly—not just for childhood immunizations. Vembu’s posts cited a non-peer-reviewed report co-authored by Andrew Wakefield, whose fraudulent 1998 Lancet paper was retracted. Yet he framed this as “brave doctors questioning dogma.” When a tech entrepreneur with no medical training publicly casts doubt on vaccine safety, it provides social proof for vaccine-hesitant individuals to justify their decisions. That skepticism bleeds into decisions about flu shots, pneumococcal vaccines, and COVID boosters—decisions that directly impact cardiovascular risk in adults.
System-level barriers. Fragmented care separates preventive services from specialty cardiac care. Vaccines are viewed as primary care’s responsibility, not cardiology’s, leading to missed opportunities. Electronic health record systems often lack integrated prompts linking cardiovascular diagnoses to vaccination recommendations. Cost and access barriers persist, particularly in populations without insurance coverage or in regions where vaccine availability is limited.
The solution requires action at all levels: standing orders allowing nursing staff to administer vaccines, co-location of vaccination services within cardiology clinics, and public health campaigns countering misinformation.
Until these changes occur, patients need to advocate for themselves. At your next cardiology appointment, ask: “Am I up to date on my flu, pneumococcal, COVID-19, and RSV vaccines?” Frame it as cardiovascular prevention, not infection control.
The evidence speaks—if we listen
The irony is stark. While a tech billionaire with no medical training commands millions of views spreading vaccine misinformation, landmark cardiovascular trials receive little public attention. The IAMI trial’s 41% reduction in cardiovascular mortality didn’t go viral. The dose-response data showing 83% protection against post-COVID cardiac events didn’t trend on X.
Misinformation drowns out evidence. And the casualties aren’t just childhood vaccination rates—they’re middle-aged adults with coronary disease who skip their flu shots, elderly patients with heart failure who never get pneumococcal vaccines, and post-MI patients who leave the hospital without the intervention that could cut their death risk nearly in half.
The conversation we’re having about vaccines is the wrong one. The conversation we should be having is in the data I’ve presented above. The question is whether cardiologists, patients, and public health systems will act on it before the next patient becomes a preventable statistic.
If you found this useful, please share it.
Evidence over opinions. Clarity over noise. That’s Signal Over Noise.
REFERENCES
Fröbert O, Götberg M, Erlinge D, et al. Influenza Vaccination After Myocardial Infarction: A Randomized, Double-Blind, Placebo-Controlled, Multicenter Trial. Circulation. 2021;144(18):1476-1484. https://doi.org/10.1161/CIRCULATIONAHA.121.057042
Warren-Gash C, Hayward AC, Hemingway H, et al. Influenza infection and risk of acute myocardial infarction in England and Wales: a CALIBER self-controlled case series study. J Infect Dis. 2012;206(11):1652-1659.
Udell JA, Zawi R, Bhatt DL, et al. Association between influenza vaccination and cardiovascular outcomes in high-risk patients: a meta-analysis. JAMA. 2013;310(16):1711-1720.
Multi-omics analysis reveals COVID-19 vaccine induced attenuation of inflammatory responses during breakthrough disease. Nat Commun. 2024;15(1):3402.
Metabolic pathways and trained immunity: Plasma metabolome predicts trained immunity responses after antituberculosis BCG vaccination. PLoS Biol. 2022;20(9):e3001765.
Netea MG, Domínguez-Andrés J, Barreiro LB, et al. Defining trained immunity and its role in health and disease. Nat Rev Immunol. 2020;20(6):375-388.
BNT162b2 or CoronaVac Vaccinations Are Associated With a Lower Risk of Myocardial Infarction and Stroke After SARS-CoV-2 Infection Among Patients With Cardiovascular Disease. J Am Heart Assoc. 2023;12(8):e029291.
The role of COVID-19 vaccines in preventing post-COVID-19 thromboembolic and cardiovascular complications. Heart. 2024;110(9):567-573.
Yadlowsky S, Patel RC, Chua K, et al. Estimation and Validation of Ratio-based Conditional Average Treatment Effects Using Observational Data. J Am Stat Assoc. 2021;116(533):335-352.
Bivalent RSV Prefusion F Protein-Based Vaccine for Preventing Cardiovascular Hospitalizations in Older Adults: A Prespecified Analysis of the DAN-RSV Trial. JAMA. 2025;333(6):512-521.
Respiratory Syncytial Virus Prefusion F Protein Vaccine Is Efficacious in Older Adults With Underlying Medical Conditions. Clin Infect Dis. 2024;78(3):e1-e12.
Simultaneous vaccination against influenza and respiratory syncytial virus in high-risk heart failure patients. Eur J Heart Fail. 2024;26(8):1755-1763.
Incidence of Myocarditis/Pericarditis Following mRNA COVID-19 Vaccination Among Children and Younger Adults in the United States. Ann Intern Med. 2022;175(12):1169-1177.
Pneumococcal and Influenza Vaccination Coverage in Patients with Heart Failure: A Systematic Review. J Clin Med. 2024;13(11):3029.
Pneumococcal and influenza vaccination rates among patients with cardiovascular disease. J Health Sci Med. 2022;5(4):1102321.
Effects of Influenza Vaccine on Mortality and Cardiovascular Outcomes in Patients With Cardiovascular Disease: A Systematic Review and Meta-Analysis. J Am Heart Assoc. 2021;10(11):e019636.