Cardiology Finally Has a Prevention Drug
Rethinking Cardiovascular Disease in the GLP-1 Era
For the better part of a century, cardiology has been an acute care specialty masquerading as a preventive one. We became exceptionally skilled at the plumbing: opening occluded arteries with stents, restarting stalled hearts with electricity. But we remained frustratingly ineffective at preventing the biological slide that leads patients into the catheterization lab in the first place.
Prevention felt like trench warfare. We won significant battles with statins in the 1970s and 1980s, effectively neutralizing LDL cholesterol. We gained ground with antihypertensives. We even cut smoking rates. Yet despite these victories, the enemy kept advancing. Obesity and type 2 diabetes continued to climb, acting as twin accelerants on the vascular system.
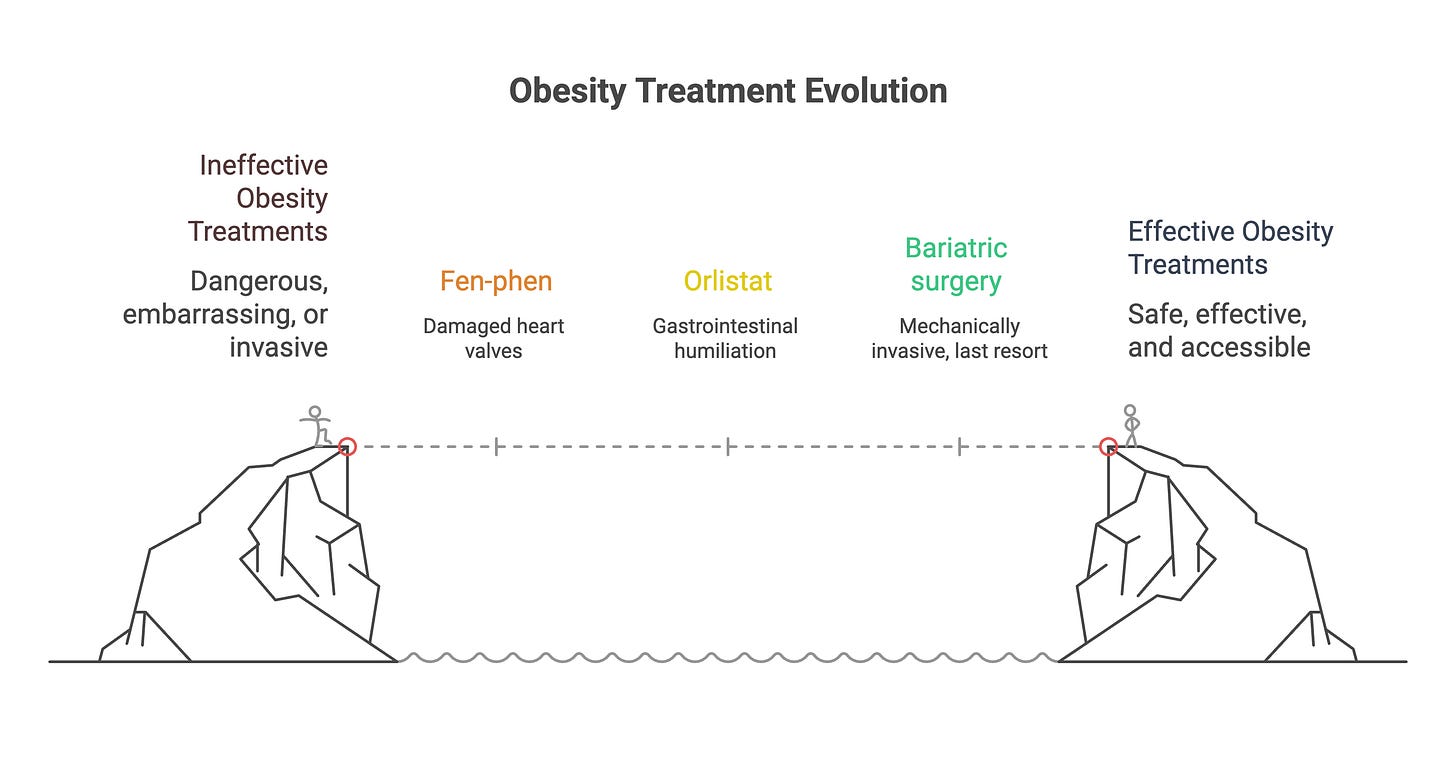
For decades, the pharmacological tools to fight obesity were either dangerous or embarrassing. Fen-phen damaged heart valves (1). Orlistat resulted in gastrointestinal humiliation. Bariatric surgery worked but was mechanically invasive and framed as a desperate last resort.
When GLP-1 receptor agonists first appeared, the medical establishment largely pigeonholed them as just another diabetes drug. They were expected to lower hemoglobin A1c, perhaps cause a little weight loss, and stay in their endocrinology lane. Then the cardiovascular outcome trials began to land, and the silo walls collapsed.
The Biology of the Whisper
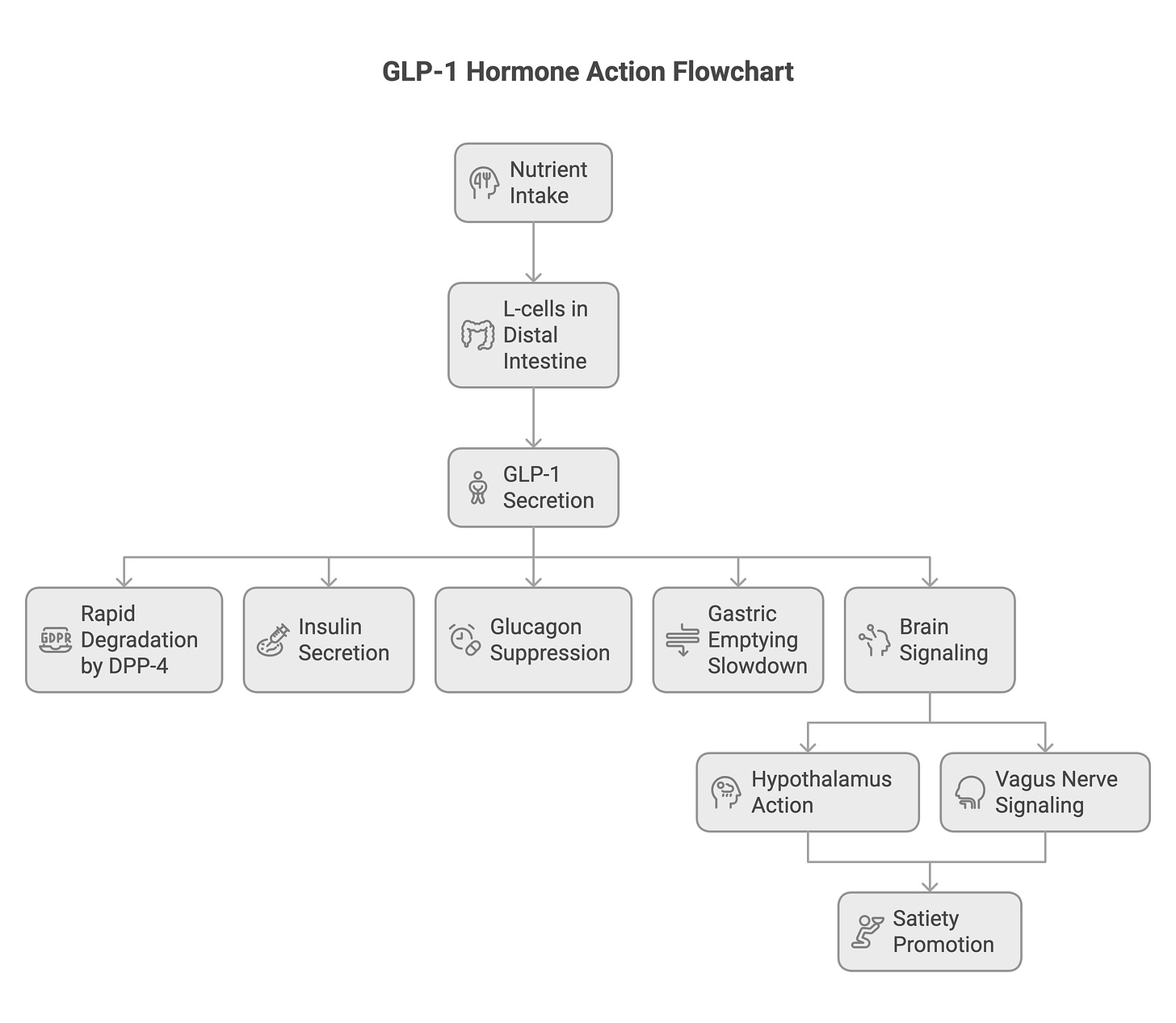
To understand why this class of drugs has upended cardiology, you need to understand the hormone itself. Glucagon-like peptide-1 (GLP-1) is an incretin hormone secreted by L-cells in the distal intestine in response to nutrient intake. In its native state, GLP-1 is ephemeral. It survives for only 1 to 2 minutes in the bloodstream before being degraded by dipeptidyl peptidase-4 (DPP-4) (2). In its natural state, GLP-1 shows up after you eat, does its job, and vanishes within minutes.
It nudges insulin secretion upward in a glucose-dependent manner, suppresses glucagon to stop the liver from overproducing sugar, and slows gastric emptying (3). Crucially, it also talks to the brain. Through direct action on the hypothalamus and indirect signaling via the vagus nerve, GLP-1 promotes satiety(4).
Scientists circumvented the rapid degradation of native GLP-1 by engineering analogues: molecules that mimic the hormone but resist enzymatic destruction, lasting for days or weeks. When these stable ligands flood the receptors, they don’t just lower blood sugar. They modulate the central appetite drive, which patients have started calling the “food noise.”(5)
This phrase went viral on social media not because it was marketing language but because it captured something people hadn’t been able to articulate before. Not hunger itself, but the constant mental chatter around eating. This was the first clue that we were dealing with something more profound than a glucose-lowering agent. We were manipulating fundamental neurobehavioral pathways.
The Data That Changed the Paradigm
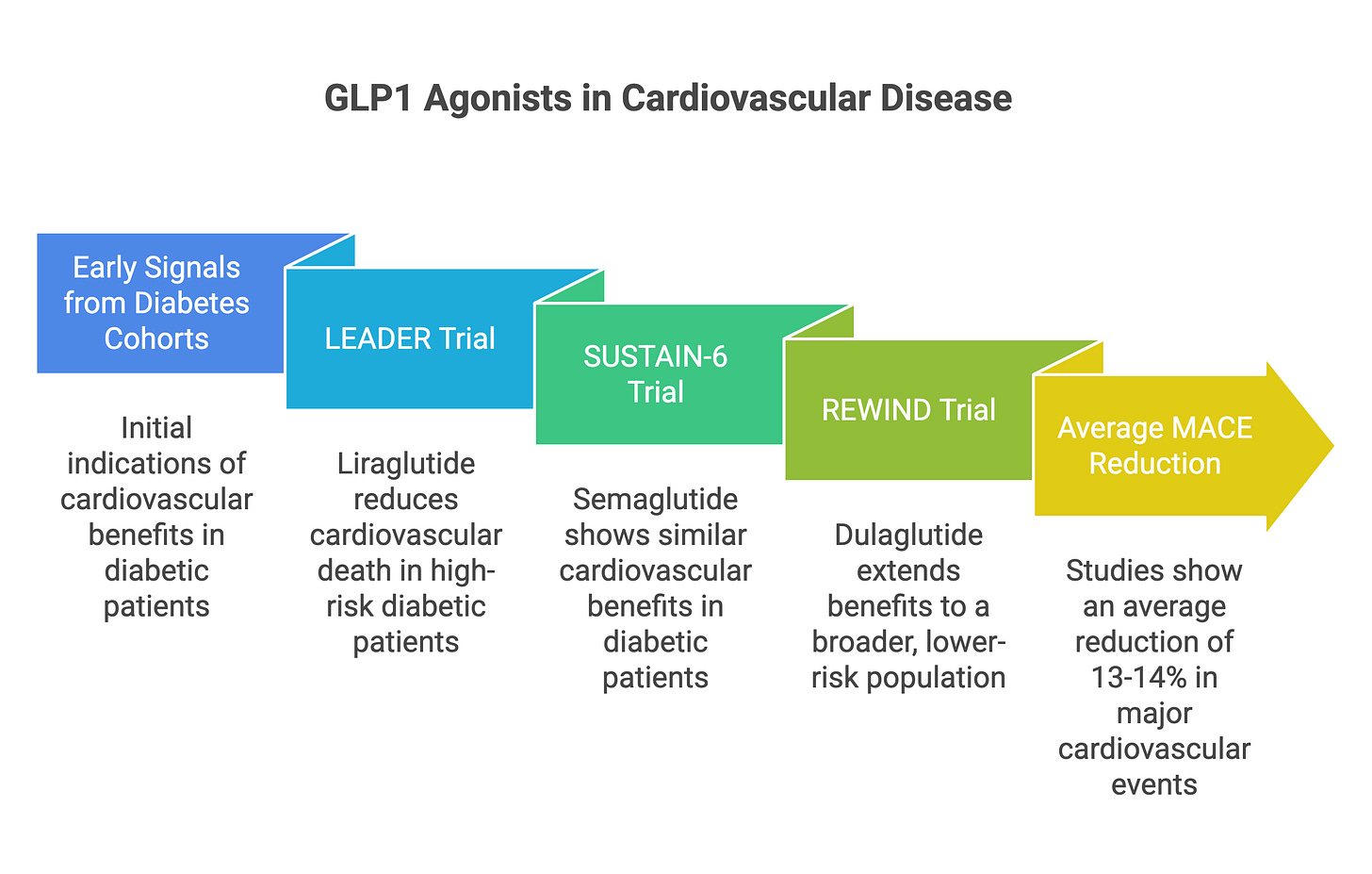
The narrative shift from “diabetes drug” to “cardiovascular drug” was built on a foundation of rigorous, large-scale randomized controlled trials. The early signals came from diabetes cohorts. The LEADER trial demonstrated that liraglutide reduced cardiovascular death in high-risk diabetic patients (6). SUSTAIN-6 showed similar benefits with semaglutide (7), and REWIND extended these findings to a broader, lower-risk population with dulaglutide (8). Across these studies, the reduction in major adverse cardiovascular events (MACE: a composite of heart attack, stroke, and cardiovascular death) averaged around 13 to 14%.
This was impressive, but it remained tethered to diabetes. The real existential shock to the system came with the SELECT trial in 2023. SELECT asked a bold question: Could semaglutide reduce heart attacks and strokes in people who were overweight or obese but did not have diabetes? The study enrolled 17,604 patients with established cardiovascular disease. The results were unequivocal. Semaglutide 2.4 mg reduced the risk of MACE by 20% (hazard ratio 0.80, 95% CI 0.72-0.90, P<0.001)(9).
This was the turning point. The drug didn’t just fix sugar; it fixed the substrate of vascular disease itself. In SELECT, patients lost approximately 10% of their body weight, but they also saw reductions in systolic blood pressure, triglycerides, and C-reactive protein (a marker of inflammation). The data suggested that treating adiposity was practically synonymous with treating the artery.
Beyond the Heart: The Multi-Organ Benefit
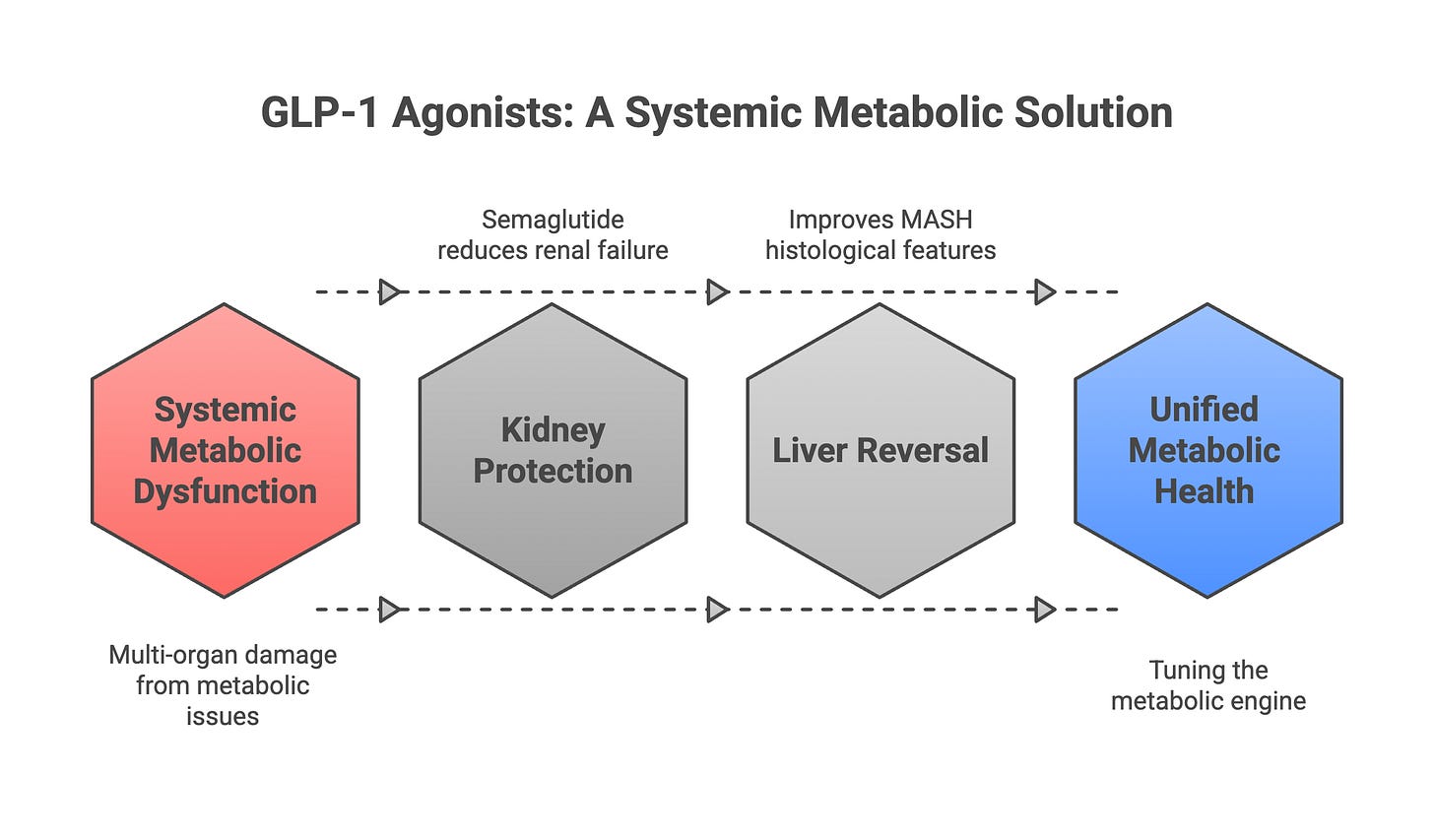
The story has since expanded beyond the cardiovascular system, revealing that metabolic dysfunction is a systemic crisis, not a localized one. The kidneys, often the first casualties of diabetes and hypertension, are also protected. The FLOW trial was stopped early for efficacy, a rarity in nephrology. It showed that semaglutide significantly reduced the progression of renal failure and death in patients with type 2 diabetes and chronic kidney disease (10).
Simultaneously, hepatologists are seeing reversals in metabolic dysfunction-associated steatohepatitis (MASH), the liver manifestation of metabolic syndrome. Semaglutide has been shown to improve histological features of MASH (11).
We are witnessing a pharmacological unification theory. Heart failure, kidney disease, fatty liver, and atherosclerosis are not separate diseases requiring separate specialists. They are different faces of the same metabolic engine. GLP-1 receptor agonists tune that engine.
The Weight Loss That Matters
The magnitude of weight loss with GLP-1 receptor agonists exceeded initial expectations. The STEP 1 trial of semaglutide 2.4 mg weekly demonstrated 14.9% weight loss at 68 weeks in patients with obesity (12). The SURMOUNT-1 trial of tirzepatide (a dual GLP-1/GIP receptor agonist) achieved 20.9% weight loss with the 15 mg dose (13), surpassing any previous pharmacotherapy for obesity.
A critical question: does cardiovascular benefit derive solely from weight loss, or do GLP-1 receptor agonists exert weight-independent cardioprotection? Several lines of evidence suggest weight-independent mechanisms contribute substantially. In cardiovascular outcome trials, Kaplan-Meier curves for MACE begin diverging within months, before substantial weight loss occurs. The remaining benefit appears attributable to direct vascular, anti-inflammatory, and potentially myocardial effects.
Numbers alone rarely move people, but weight loss is not vanity in cardiology. It’s hemodynamics. A story of a patient discovering they can climb a flight of stairs without stopping conveys more truth than any hazard ratio. That is the cardiovascular math embodied.
Addressing the Noise: Safety and Controversy
No therapy this potent comes without controversy, and the cultural explosion around “Ozempic” has generated significant noise. We must distinguish between physiological risks and social media panic.
Gastrointestinal symptoms remain the most common cause of discontinuation, occurring in 20 to 40% of patients depending on dose. These symptoms are usually mild to moderate, occur early in therapy, and diminish over time. Slow dose titration substantially reduces their frequency and severity. More serious concerns, such as gastroparesis (stomach paralysis), have appeared in case reports. While rare, these underscore the need for careful patient selection and dose escalation.
The pancreatitis debate continues. While observational signals exist, meta-analyses of randomized trials have not confirmed a definitive causal link. Meta-analyses do confirm increased risk of gallbladder disease (14) likely related to rapid weight loss.
Critics frequently cite the loss of lean muscle mass as a fatal flaw. Physiologically, this is expected. Any rapid weight loss, whether surgical or dietary, involves catabolism of both fat and lean tissue. The solution is not to abandon the drug but to pair it with resistance training and adequate protein intake, standard advice for any weight management intervention.
Perhaps the most stubborn criticism is that “the weight comes back when you stop.” Withdrawal studies confirm this: patients regain substantial weight after cessation(15). This is often framed as a failure of the drug. In reality, it is proof of the disease. We don’t criticize antihypertensives because blood pressure rises when pills are stopped. We don’t mock statins because cholesterol rebounds upon discontinuation. Obesity is a chronic, relapsing neuro-metabolic disease. It requires chronic treatment.
Thyroid C-cell tumors appeared in rodent studies, leading to boxed warnings, but human evidence has not confirmed this risk after nearly two decades of use (16,17)
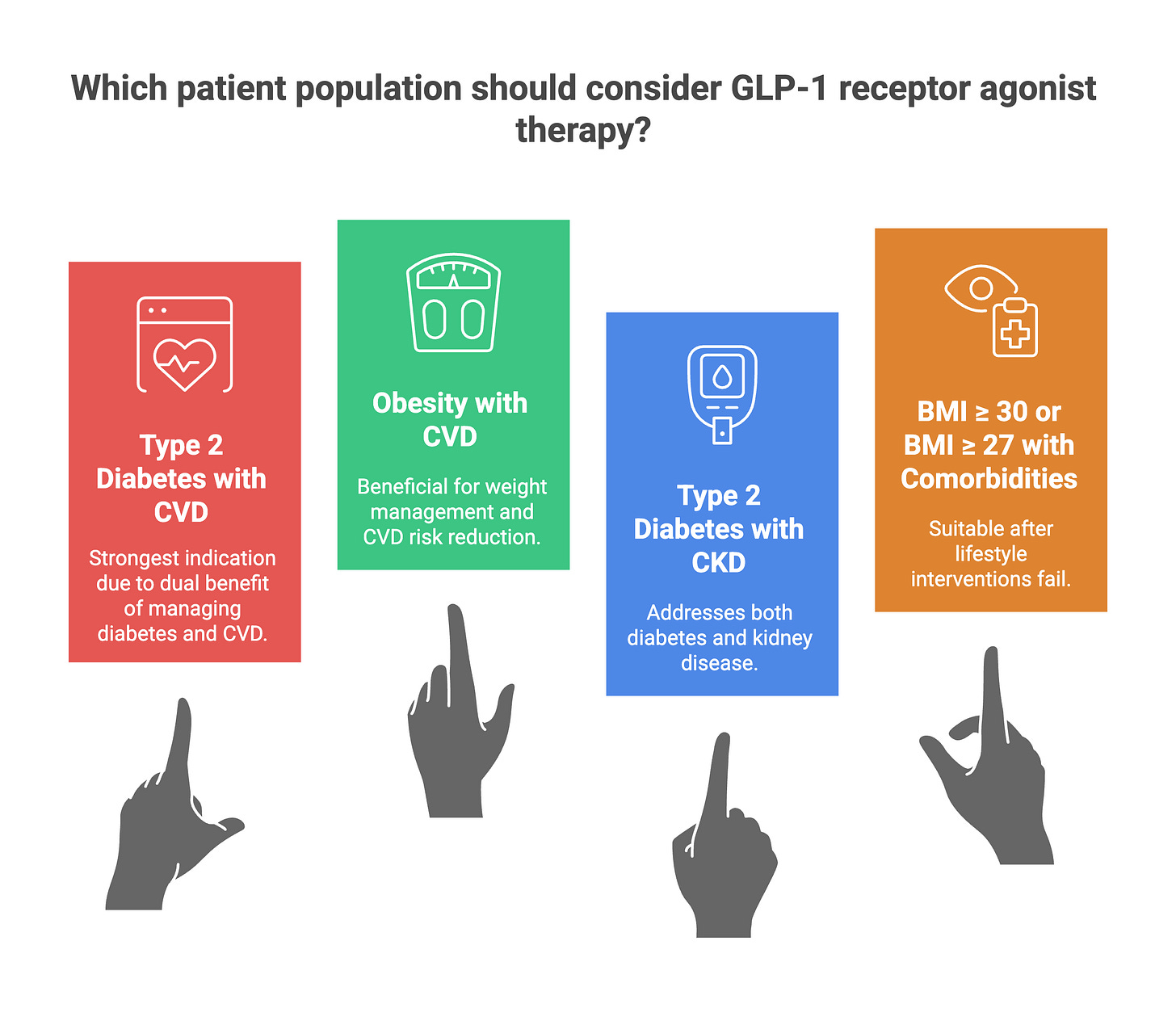
Who Should Consider Therapy
Current guidelines provide clear direction. The 2025 ADA Standards of Care and 2023 ESC guidelines give strong recommendations for GLP-1 receptor agonists in patients with type 2 diabetes and established atherosclerotic cardiovascular disease. For cardiologists and clinicians, several patient populations warrant consideration:
Type 2 diabetes with established cardiovascular disease (the strongest indication)
Obesity with established cardiovascular disease regardless of diabetes status (the SELECT population)
Type 2 diabetes with chronic kidney disease (the FLOW population)
BMI 30 kg/m² or greater or BMI 27 kg/m² or greater with comorbidities after lifestyle interventions have failed
The Horizon: Science Fiction Becoming Science Fact
We are currently only at the beginning of the incretin era. The pipeline is moving from single agonists to multi-receptor engineering. Tirzepatide has already demonstrated weight loss exceeding 20% in the SURMOUNT-1 trial, surpassing the efficacy of semaglutide(13).
Retatrutide, a “triple G” agonist (GLP-1, GIP, and glucagon), showed weight loss of 24.2% at 48 weeks with the 12 mg dose in a phase 2 trial (18). This level of efficacy was previously the exclusive domain of invasive bariatric surgery.
Oral formulations promise to break the barrier of injectability, potentially democratizing access if costs can be managed. This is where medicine starts to feel like speculative fiction, except the trials are real(19,20).
Why Lifestyle Still Matters
GLP-1 receptor agonists target the biological drivers of weight, but lifestyle addresses muscle mass, VO₂ max, sleep, mood, and metabolic resilience. All major guidelines treat medication as adjunctive to behavioral intervention. These drugs don’t replace lifestyle. They make lifestyle possible again for people whose biology has fought them for decades.
Where This Leaves Us
The arrival of effective GLP-1 therapies forces a rare moment of clarity in medicine. For years, we treated the downstream consequences of metabolic dysfunction: stenting the blocked artery, dialyzing the failed kidney, transplanting the cirrhotic liver, while the root cause remained untreated. The science now stands firm. We have clear evidence of fewer heart attacks, fewer strokes, preserved kidney function, and profound weight loss.
There will be challenges ahead. Cost poses a massive ethical dilemma, creating a rift where the wealthy access health and the vulnerable access advice. Monthly costs of $900 to $1,500 for brand-name GLP-1 receptor agonists create barriers for patients and healthcare systems. The long-term effects of decades-long use remain to be fully mapped.
But for the first time in cardiology’s history, we are not just waiting in the trenches for the enemy to attack. We are finally dismantling the engine of the disease itself. The GLP-1 era forces a conceptual shift: obesity, diabetes, cardiovascular disease, liver disease, and kidney disease are different manifestations of the same underlying metabolic engine. For the first time, we have drugs that tune that engine directly.
The cardiovascular community must take ownership of GLP-1 receptor agonist prescribing for our patients. Integration into cardiology practice, development of cardiometabolic clinics, and advocacy for appropriate coverage represent necessary next steps in translating this powerful evidence base into improved patient outcomes. The fundamental question is no longer whether GLP-1 receptor agonists reduce cardiovascular events (the evidence is definitive) but rather how we ensure that patients who would benefit actually receive them.
If you found this useful, please share it.
Evidence over opinions. Clarity over noise. That’s Signal Over Noise.
References
1. Connolly HM, Crary JL, McGoon MD, Hensrud DD, Edwards BS, Edwards WD, et al. Valvular heart disease associated with fenfluramine-phentermine. N Engl J Med [Internet]. 1997 Aug 28;337(9):581–8. Available from: https://doi.org/10.1056/NEJM199708283370901
2. Singh AK. Dipeptidyl peptidase-4 inhibitors: Novel mechanism of actions. Indian J Endocrinol Metab [Internet]. 2014 Nov;18(6):753–9. Available from: http://dx.doi.org/10.4103/2230-8210.141319
3. Holst JJ. The physiology of glucagon-like peptide 1. Physiol Rev [Internet]. 2007 Oct;87(4):1409–39. Available from: http://dx.doi.org/10.1152/physrev.00034.2006
4. van Bloemendaal L, Ten Kulve JS, la Fleur SE, Ijzerman RG, Diamant M. Effects of glucagon-like peptide 1 on appetite and body weight: focus on the CNS. J Endocrinol [Internet]. 2014 Apr;221(1):T1–16. Available from: http://dx.doi.org/10.1530/JOE-13-0414
5. Naveed M, Perez C, Ahmad E, Russell L, Lees Z, Maybury C. GLP-1 medication and weight loss: Barriers and motivators among 1659 participants managed in a virtual setting. Diabetes Obes Metab [Internet]. 2025 July;27(7):3780–8. Available from: http://dx.doi.org/10.1111/dom.16405
6. Marso SP, Daniels GH, Brown-Frandsen K, Kristensen P, Mann JFE, Nauck MA, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med [Internet]. 2016 July 28;375(4):311–22. Available from: https://doi.org/10.1056/NEJMoa1603827
7. Marso SP, Bain SC, Consoli A, Eliaschewitz FG, Jódar E, Leiter LA, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med [Internet]. 2016 Nov 10;375(19):1834–44. Available from: http://dx.doi.org/10.1056/NEJMoa1607141
8. Gerstein HC, Colhoun HM, Dagenais GR, Diaz R, Lakshmanan M, Pais P, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet [Internet]. 2019 July 13;394(10193):121–30. Available from: https://doi.org/10.1016/S0140-6736(19)31149-3
9. Lincoff AM, Brown-Frandsen K, Colhoun HM, Deanfield J, Emerson SS, Esbjerg S, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med [Internet]. 2023 Dec 14;389(24):2221–32. Available from: https://doi.org/10.1056/NEJMoa2307563
10. Perkovic V, Tuttle KR, Rossing P, Mahaffey KW, Mann JFE, Bakris G, et al. Effects of semaglutide on chronic kidney disease in patients with type 2 diabetes. N Engl J Med [Internet]. 2024 July 11;391(2):109–21. Available from: https://doi.org/10.1056/NEJMoa2403347
11. Newsome PN, Buchholtz K, Cusi K, Linder M, Okanoue T, Ratziu V, et al. A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis. N Engl J Med [Internet]. 2021 Mar 25;384(12):1113–24. Available from: https://doi.org/10.1056/NEJMoa2028395
12. Wilding JPH, Batterham RL, Calanna S, Davies M, Van Gaal LF, Lingvay I, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med [Internet]. 2021 Mar 18;384(11):989–1002. Available from: https://doi.org/10.1056/NEJMoa2032183
13. Jastreboff AM, Aronne LJ, Ahmad NN, Wharton S, Connery L, Alves B, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med [Internet]. 2022 July 21;387(3):205–16. Available from: https://doi.org/10.1056/NEJMoa2206038
14. Chiang CH, Jaroenlapnopparat A, Colak SC, Yu CC, Xanthavanij N, Wang TH, et al. Glucagon-like peptide-1 receptor agonists and gastrointestinal adverse events: A systematic review and meta-analysis. Gastroenterology [Internet]. 2025 Nov;169(6):1268–81. Available from: http://dx.doi.org/10.1053/j.gastro.2025.06.003
15. Wilding JPH, Batterham RL, Davies M, Van Gaal LF, Kandler K, Konakli K, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes Obes Metab [Internet]. 2022 Aug;24(8):1553–64. Available from: https://doi.org/10.1111/dom.14725
16. Chiu WY, Shih SR, Tseng CH. A review on the association between glucagon-like peptide-1 receptor agonists and thyroid cancer. Exp Diabetes Res [Internet]. 2012 May 28;2012:924168. Available from: http://dx.doi.org/10.1155/2012/924168
17. Espinosa De Ycaza AE, Brito JP, McCoy RG, Shao H, Singh Ospina N. Glucagon-like peptide-1 receptor agonists and thyroid cancer: A narrative review. Thyroid [Internet]. 2024 Apr;34(4):403–18. Available from: http://dx.doi.org/10.1089/thy.2023.0530
18. Jastreboff AM, Kaplan LM, Frías JP, Wu Q, Du Y, Gurbuz S, et al. Triple-hormone-receptor agonist retatrutide for obesity - A phase 2 trial. N Engl J Med [Internet]. 2023 Aug 10;389(6):514–26. Available from: https://doi.org/10.1056/NEJMoa2301972
19. Wharton S, Blevins T, Connery L, Rosenstock J, Raha S, Liu R, et al. Daily oral GLP-1 receptor agonist orforglipron for adults with obesity. N Engl J Med [Internet]. 2023 Sept 7;389(10):877–88. Available from: http://dx.doi.org/10.1056/NEJMoa2302392
20. Drucker DJ. Efficacy and safety of GLP-1 medicines for type 2 diabetes and obesity. Diabetes Care [Internet]. 2024 Nov 1;47(11):1873–88. Available from: https://dx.doi.org/10.2337/dci24-0003